Text Solution
Verified by Experts
Topper's Solved these Questions
ORGANIC COMPOUNDS CONTAINING NITROGEN
VIKRAM PUBLICATION ( ANDHRA PUBLICATION)|Exercise INTEXT QUESTIONS|14 VideosORGANIC COMPOUNDS CONTAINING NITROGEN
VIKRAM PUBLICATION ( ANDHRA PUBLICATION)|Exercise DAM SURE (VSAQ - 2 Marks)|16 VideosORGANIC COMPOUNDS CONTAINING NITROGEN
VIKRAM PUBLICATION ( ANDHRA PUBLICATION)|Exercise SHORT ANSWER QUESTIONS|19 VideosORGANIC COMPOUNDS CONTAINING C,H AND O
VIKRAM PUBLICATION ( ANDHRA PUBLICATION)|Exercise INTEXT QUESTIONS|54 VideosP-BLOCK ELEMENTS
VIKRAM PUBLICATION ( ANDHRA PUBLICATION)|Exercise INTEXT QUESTIONS|30 Videos
Similar Questions
Explore conceptually related problems
VIKRAM PUBLICATION ( ANDHRA PUBLICATION)-ORGANIC COMPOUNDS CONTAINING NITROGEN-LONG ANSWER QUESTIONS
- Write the product formed when benzyl chloride is reacted with ammonia ...
Text Solution
|
- Identify the amide and cyanide which on reduction with appropriate red...
Text Solution
|
- Write the mechanism of Hoffmann bromamide reaction.
Text Solution
|
- How do you make the following convertions ? Chlorophenylmethane to p...
Text Solution
|
- How do you make the following convertions ? Chlorophenylmethane to 2...
Text Solution
|
- Identify the starting amide which gives p - methyl aniline on reaction...
Text Solution
|
- Explain why the order of basicity for methyl amine, N, N - dimethyl am...
Text Solution
|
- Write the equations involved in the reaction of Nitrous acid with Ethy...
Text Solution
|
- Explain with equations how methylamine, N, N - dimethylamine and N, N,...
Text Solution
|
- Explain why aniline in strong acidic medium gives a mixture of Nitro a...
Text Solution
|
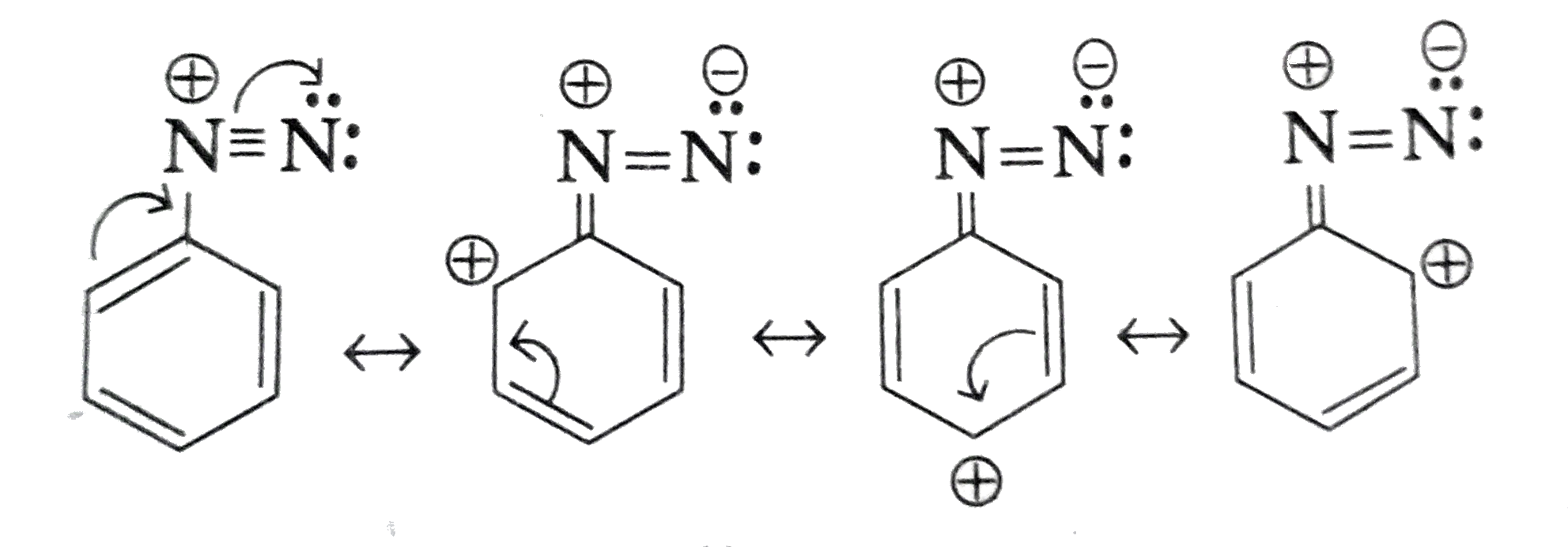
- Account for the stability of aromatic diazonium ions when compared to ...
Text Solution
|
- Write the equations showing the conversion of aniline diazoniumchlorid...
Text Solution
|
- Complete the following conversions : Aniline to Fluorobenzene
Text Solution
|
- Complete the following conversions : Aniline to Cyanobenzene
Text Solution
|
- Complete the following conversions : Aniline to Benzene
Text Solution
|
- Complete the following conversions : Aniline to Phenol
Text Solution
|
- Explain the following name reactions : Sandmeyer reaction
Text Solution
|
- Explain the following name reactions : Gatterman reaction
Text Solution
|
- Write the steps involved in the coupling of Benzene diazoniumchloride ...
Text Solution
|
- Write the equations involved in the conversion of acetamide and propan...
Text Solution
|