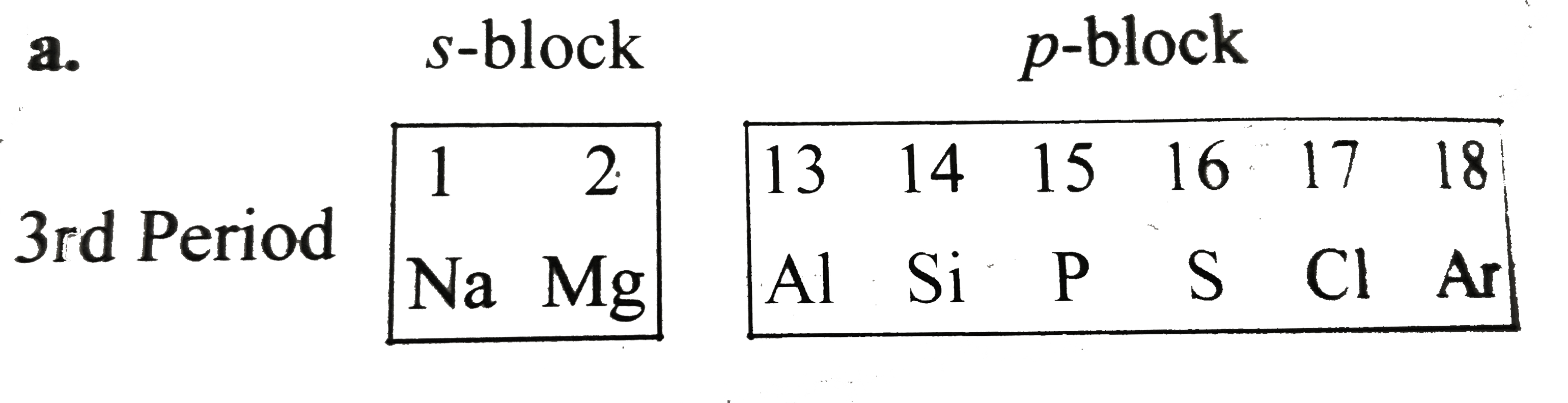A
B
C
D
Text Solution
Verified by Experts
The correct Answer is:
Topper's Solved these Questions
PERIODIC CLASSIFICATION OF ELEMENTS AND GENERAL INORGANIC CHEMISTRY
CENGAGE CHEMISTRY|Exercise Exercises (Singlecorrect) Atomic And Ionic Radii|15 VideosPERIODIC CLASSIFICATION OF ELEMENTS AND GENERAL INORGANIC CHEMISTRY
CENGAGE CHEMISTRY|Exercise Exercises (Single Correct) Ionisation Energy (Ie)|31 VideosPERIODIC CLASSIFICATION OF ELEMENTS AND GENERAL INORGANIC CHEMISTRY
CENGAGE CHEMISTRY|Exercise Exercises (Multiple Correct) Miscellaneous|21 VideosP-BLOCK GROUP 14 - CARBON FAMILY
CENGAGE CHEMISTRY|Exercise Exercises Archives (Subjective)|9 VideosPURIFICATION OF ORGANIC COMPOUNDS AND QUALITATIVE AND QUANTITATIVE ANALYSIS
CENGAGE CHEMISTRY|Exercise Assertion Reasoning Type|5 Videos
Similar Questions
Explore conceptually related problems
CENGAGE CHEMISTRY-PERIODIC CLASSIFICATION OF ELEMENTS AND GENERAL INORGANIC CHEMISTRY-Exercises (Singlecorrect) General Electronic Configuration And Periodicity
- The statement that is false regarding the long form the periodic tabl...
Text Solution
|
- In the periodic table, going down in fluorine group
Text Solution
|
- In the long form of periodic table all the non-metals are placed under
Text Solution
|
- Alkali metals in each period have
Text Solution
|
- Which one pair of atoms or ions will have same configuration ?
Text Solution
|
- In the modern periodic table, the period indicates the value of
Text Solution
|
- Which of the following does not reflect periodicity of elements ?
Text Solution
|
- The 3rd period of the periodic table contains
Text Solution
|
- Which of the following set
Text Solution
|
- Which of the following belongs to the category of transition metal?
Text Solution
|
- Without looking at the periodic table, select the elements belonging t...
Text Solution
|
- The elements of same group of the periodic table have
Text Solution
|
- The elements which are characterised by the outer shell configuration ...
Text Solution
|
- The outer most electronic configuration of transition elements is
Text Solution
|
- An element with atomic number 20 will be placed in which period of the...
Text Solution
|
- The statement that is not correct for periodic classification of eleme...
Text Solution
|
- In the modern periodic table, elements are arranged in
Text Solution
|
- Name of the heaviest naturally occurring element.
Text Solution
|
- The screening effect of d-electron is
Text Solution
|
- Which of the following represents the electronic configuration of the ...
Text Solution
|