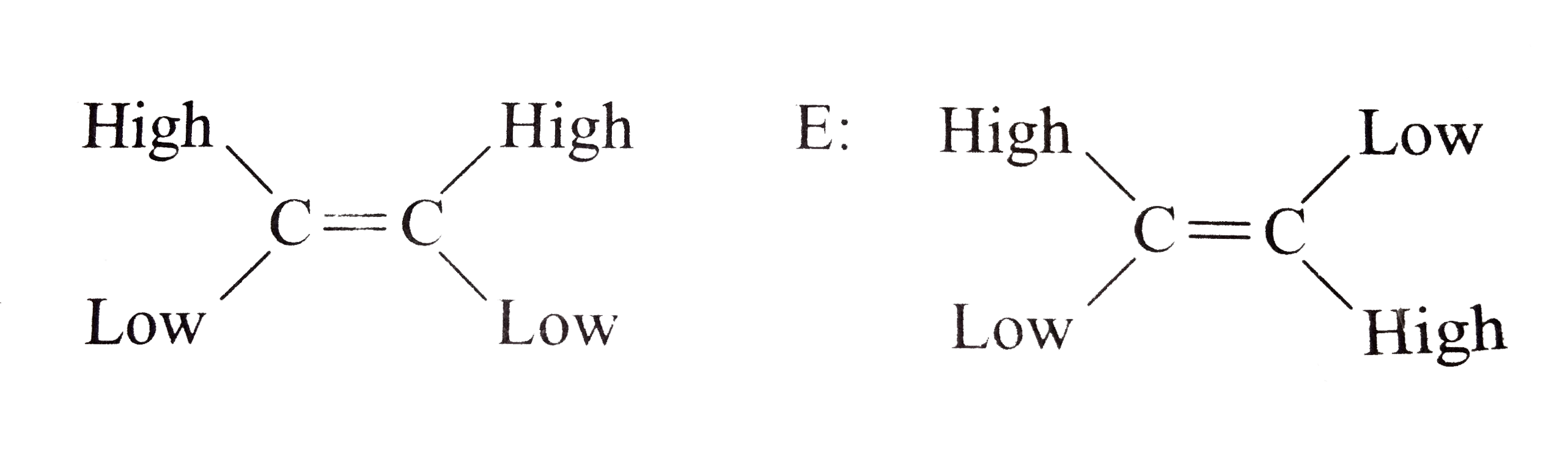Text Solution
Verified by Experts
Topper's Solved these Questions
Similar Questions
Explore conceptually related problems
R SHARMA-ALKENES-Archives
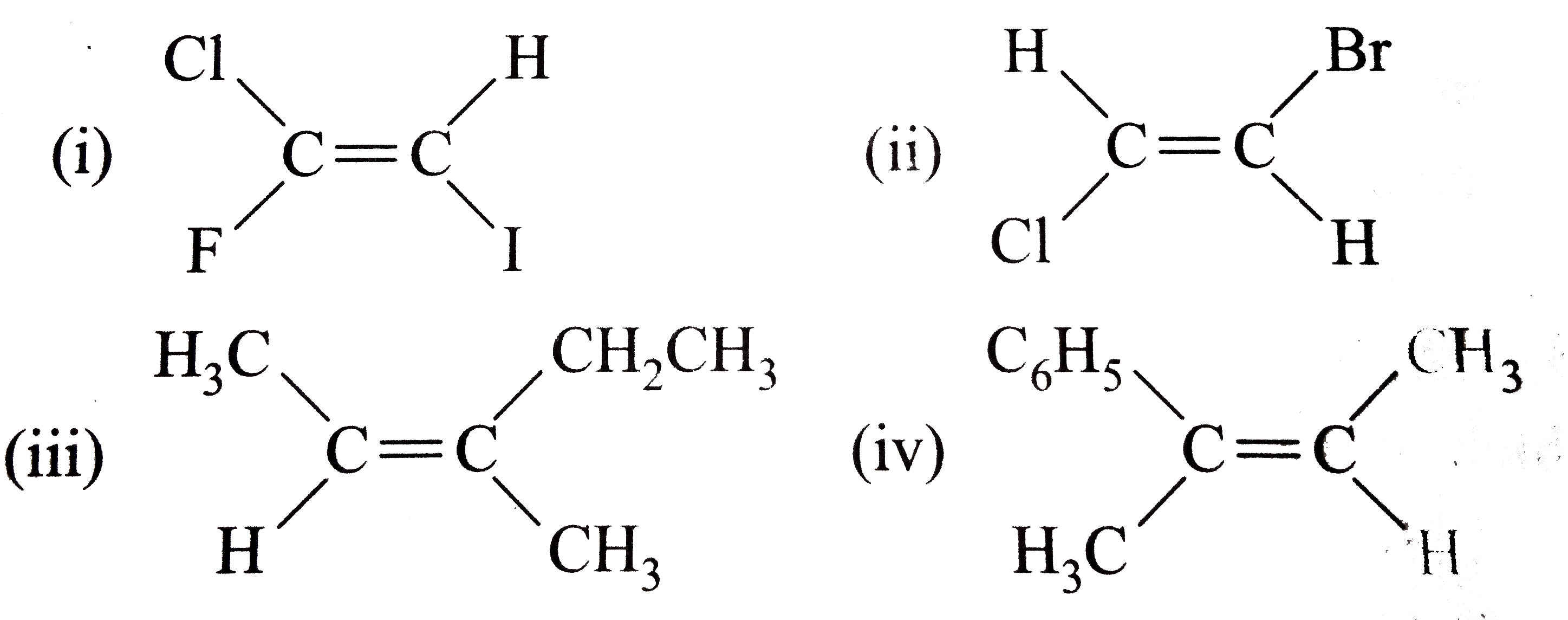
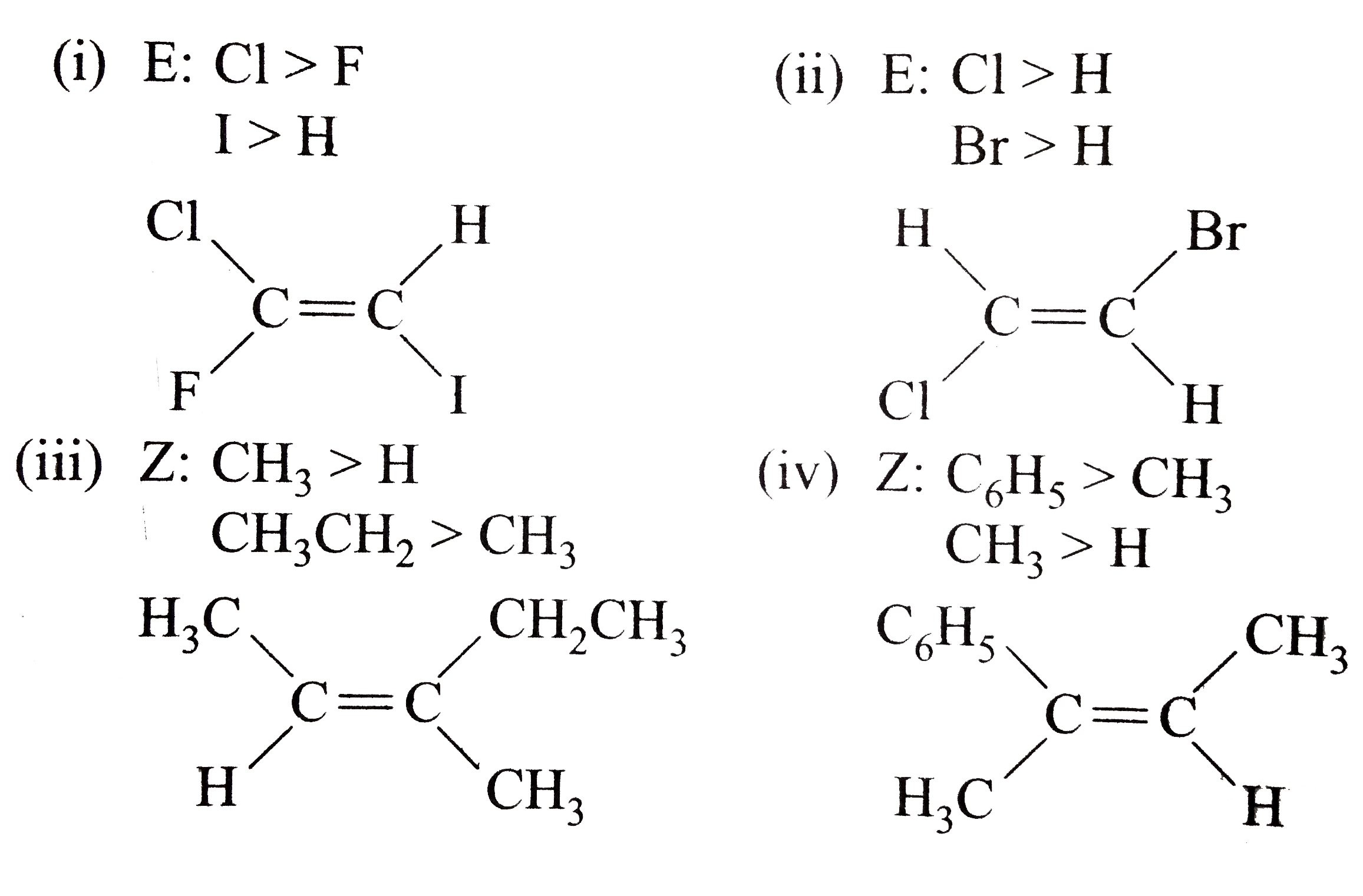
- Classify the following alkenes and their derivatives as Z or E (i) ...
Text Solution
|
- In the reaction below, X is Neopentyl alcohol overset(H(2)SO(4))rarr...
Text Solution
|
- CH(3)underset(CH(3))underset(|)CHCH=CH(2)overset(HBr)rarrA A (predom...
Text Solution
|
- Which of the following compounds with molecular formula C(5)H(10) yiel...
Text Solution
|
- The major product formed when 3,3-dimethylbutan-2-ol is heated with co...
Text Solution
|
- Oxidation of an alkene (X) gives a diol. Further oxidation gives a dik...
Text Solution
|
- The number of sigma and pi- bonds in alkyl isocyanide are
Text Solution
|
- One mole of an unsaturated hydrocarbon on ozonolysis gives one mole ea...
Text Solution
|
- Relative stabilities of various alkenes represented as R(2)C=CR(2), R(...
Text Solution
|
- 3-Phenylpropene on reaction with HBr gives (as major product)
Text Solution
|
- Which of the following alkenes will react fastest with H(2) under cata...
Text Solution
|
- Reaction of HBr with propane in the presence of peroxide gives
Text Solution
|
- The compound CH(3)-overset(CH(3))overset(|)C=CH-CH(3) on reaction wi...
Text Solution
|
- Prop-1-ol can be prepared from propene
Text Solution
|
- The reaction of HBr with CH(3)underset(CH(3))underset(|)C=CH(2) in the...
Text Solution
|
- Hydrolysis of ozonide of but-1-ene gives
Text Solution
|
- Monomer of [-underset(CH(3))underset(|)overset(CH(3))overset(|)C-CH(2)...
Text Solution
|
- Indicate the organic structure for the product expected when 2-methylp...
Text Solution
|
- In the preparation of alkene from alcohol using Al(2)O(3), which is ef...
Text Solution
|
- Which alkene on ozonolysis gives CH(3)CH(2)CHO and CH(3)COCH(3)?
Text Solution
|
- Ozonolysis of C(7)H(14) gace 2-methylpentan-3-one. The alkene is
Text Solution
|