A
B
C
D
Text Solution
Verified by Experts
The correct Answer is:
Topper's Solved these Questions
Similar Questions
Explore conceptually related problems
ALLEN-ALKYL AND ARYL HALIDE-EXERCISE
- The reaction proceeds by the mechanism
Text Solution
|
- 1,3-Dibromopropane reacts with metallic zinc to form
Text Solution
|
- Consider the following reaction sequence, CH(3)C-= CH overset(aq.H(2...
Text Solution
|
- Which of the following has highest dipole moment:
Text Solution
|
- In S(N^(1)) the first step involves the formation of
Text Solution
|
- To form alkane isonitrile, alkyl halide is reacted with:
Text Solution
|
- Which one of the following compounds most radily undergoes substitutio...
Text Solution
|
- (sec-Butyl bromide) undergoes alkaline hydrolysis by :
Text Solution
|
- Grignard reagent can be prepared by
Text Solution
|
- Most stable carbocation formed from (CH(3))(3)C-Br,(C(6)H(5))(3)CBr,(C...
Text Solution
|
- For the reaction
Text Solution
|
- The products of reaction of alcoholic silver nitrite with enthyl bromi...
Text Solution
|
- The reaction , CH(3)Br +OH^(-)rarrCH(3)OH+Br^(-) obeys the mechanism
Text Solution
|
- Ethylidene chloride can be prepared by the reaction of HCI and
Text Solution
|
- 1-phenyl-2-chloropropane on treating with alc. KOH gives mainly
Text Solution
|
- Grignard reagent is obtained when magnesium is treated with
Text Solution
|
- Ethylene reacts bromine to form-
Text Solution
|
- C(2)H(4) overset(Br(2))rarr X overset(KCN)rarr Y, Y is
Text Solution
|
- Reactivity order of halides of dehydrohalogenation is
Text Solution
|
- Which of the following is least reactive in a nucleophilic substiution...
Text Solution
|
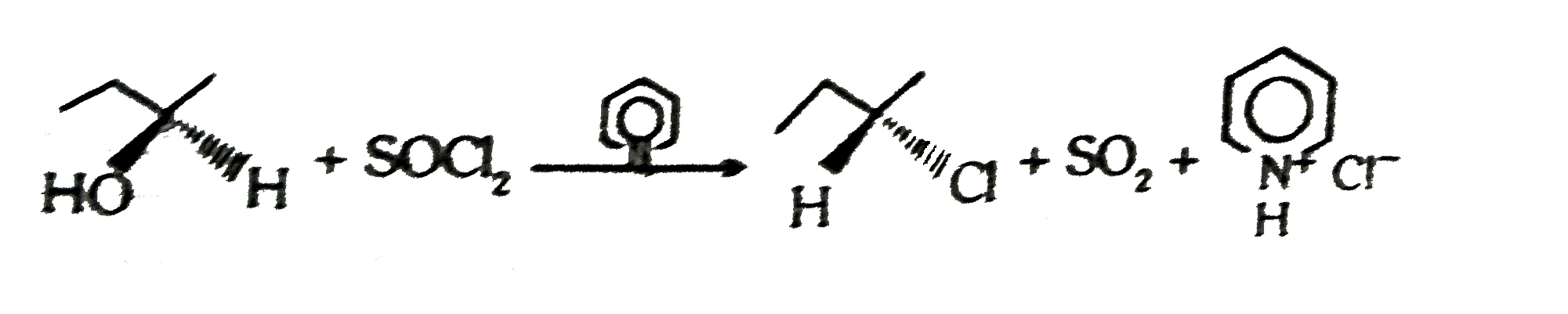 proceeds by the mechanism
proceeds by the mechanism