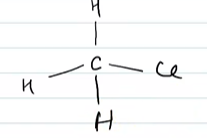
Text Solution
Verified by Experts
Topper's Solved these Questions
Similar Questions
Explore conceptually related problems
NCERT-CARBON AND ITS COMPOUNDS -Exercise
- A mixture of oxygen and ethyne is burnt for welding. Can you tell why ...
Text Solution
|
- How would you distinguish experimentally between an alcohol and a carb...
Text Solution
|
- What are oxidising agents?
Text Solution
|
- Would you be able to check if water is hard by using a detergent?
Text Solution
|
- People use a variety of methods to wash clothes. Usually after adding ...
Text Solution
|
- Ethane, with the molecular formula C(2)H(6) has
Text Solution
|
- Butanone is a four-carbon compound with the functional group
Text Solution
|
- While cooking, if the bottom of the vessel is getting blackened on the...
Text Solution
|
- Explain the nature of the covalent bond using the bond formation in CH...
Text Solution
|
- Draw the electron dot structures for (i) ethanoic acid (ii) H(2)S (i...
Text Solution
|
- What is a homologous series ? Explain with an example.
Text Solution
|
- How can ethanol and ethanoic acid be differentiated on the basis of th...
Text Solution
|
- Why does micelle formation take place when soap is added to water? Wil...
Text Solution
|
- Why are carbon and its compounds used as fuels for most applications?
Text Solution
|
- Explain the formation of scum when hard water is treated with soap.
Text Solution
|
- What change will you observe if you test soap with litmus paper (red a...
Text Solution
|
- What is hydrogenation? What is its industrial application?
Text Solution
|
- Which of the following hydrocarbons undergo addition reactions: C(2)H...
Text Solution
|
- Give a test that can be used to differentiate chemically between butte...
Text Solution
|
- Explain the mechanism of the cleansing action of soaps.
Text Solution
|