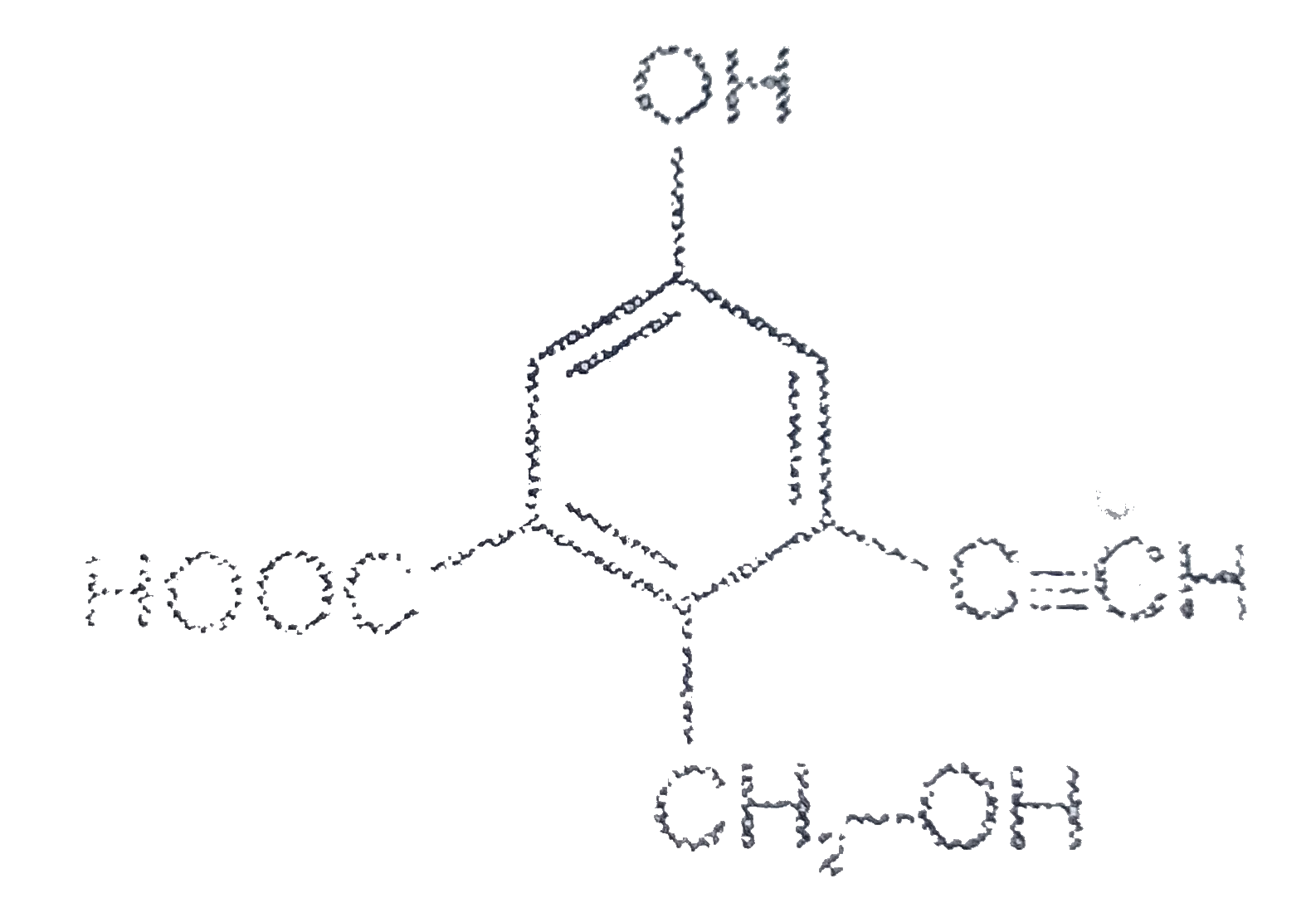
Text Solution
Verified by Experts
The correct Answer is:
Topper's Solved these Questions
STRUCTURAL IDENTIFICATION
RESONANCE|Exercise Exercise-1 (Part-II)|37 VideosSTRUCTURAL IDENTIFICATION
RESONANCE|Exercise Exercise - 2 (Part-I)|11 VideosSTRUCTURAL IDENTIFICATION
RESONANCE|Exercise Advanced level Problems (Part-III)|12 VideosSTEREOISOMERISM
RESONANCE|Exercise EXERCISE (PART III : PRACTICE TEST-2 (IIT-JEE (ADVANCED PATTERN))|22 VideosSURFACE CHEMISTRY
RESONANCE|Exercise Section - 5|1 Videos
Similar Questions
Explore conceptually related problems
RESONANCE-STRUCTURAL IDENTIFICATION-Exercise-1 (Part-I)
- How many isomeric alkyne on hydrogenation gives 3,3-Dimethylhexane.
Text Solution
|
- A cycloalkane having molecular mass 84 and four secondary carbon atoms...
Text Solution
|
- Number of monocloro structural isomers of : (i) , overset(Cl(2)//h v...
Text Solution
|
- Write the product of following reactions : (a) CH(3)-overset(H(3)C)o...
Text Solution
|
- (i) P("hydrocarbon")underset(H(2)O)overset(O(3)//Zn)rarr , , +CH(2) =...
Text Solution
|
- Write the product of following reactions : (i) CH(3)-C-=C-CH=CH(2) o...
Text Solution
|
- No. of moles of H(2)O gas evolved when one mole of the following comp...
Text Solution
|
- Molecular formula C(4)H(6) have two position isomers A and B. Both A a...
Text Solution
|
- Write suitable reagent to distiguish the following compounds.
Text Solution
|
- Identify the structure of X :
Text Solution
|
- A compound X (C(5)H(10)O) reacts with 2,4-DNP but does not give silver...
Text Solution
|
- Which of the following compounds will not give positive iodoform test....
Text Solution
|
- Identify the structure of P :
Text Solution
|
- Which of the following compound will not give positive test with NaHCO...
Text Solution
|
- Molecular formula C(3)H(6)O(2) have two structures A & B. Structure A ...
Text Solution
|
- A symmetrical organic compound of C(4)H(11)N give yellow oily layer on...
Text Solution
|
- Identify the structure of amine.
Text Solution
|
- When Lassiange extract of Methylamine react with FeSO(4)//"dilute" H(2...
Text Solution
|
- Explain the reason for the fusion of an organic compound witt metallic...
Text Solution
|
- What will happen during lassaigne's test for nitrogen if the compound ...
Text Solution
|