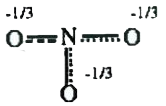
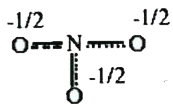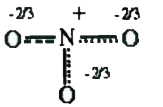A
B
C
D
Text Solution
Verified by Experts
The correct Answer is:
Topper's Solved these Questions
CHEMICAL BONDING
AAKASH SERIES|Exercise OBJECTIVE EXERCISE -2(HYDROGEN BOND )|17 VideosCHEMICAL BONDING
AAKASH SERIES|Exercise OBJECTIVE EXERCISE -2(DIPOLE MOMENT )|22 VideosCHEMICAL BONDING
AAKASH SERIES|Exercise OBJECTIVE EXERCISE -2(HYBRIDISATION AND VSEPR THEORY )|27 VideosATOMIC STRUCTURE
AAKASH SERIES|Exercise OBJECTIVE EXERCISE - 3 (RECENT AIPMT/NEET QUESTIONS)|20 VideosCHEMICAL EQUILIBRIUM
AAKASH SERIES|Exercise QUESTIONS FOR DESCRIPTIVE ANSWERS|30 Videos
Similar Questions
Explore conceptually related problems
AAKASH SERIES-CHEMICAL BONDING-OBJECTIVE EXERCISE -2(MOLECULAR ORBITAL THEORY )
- When N(2) goes to N(2)^(+), then N-N bond distance ……….and when O(2) g...
Text Solution
|
- The total number of antibonding electrons in nitrogen and oxygen molec...
Text Solution
|
- The correct order of N-O bond length in No, NO(2)^(-) and NO(3)^(-) wi...
Text Solution
|
- The Cl-O bond order in perchlorate ion
Text Solution
|
- Which combination of atomic orbitals is not allowed according to MO th...
Text Solution
|
- Arrange the following in increasing order of their bond order: O(2),O(...
Text Solution
|
- The wave function of a molecular orbital formed by reinforce of wave f...
Text Solution
|
- Which of the following orders regarding the bond order is correct?
Text Solution
|
- A bonding molecular orbital is produced by
Text Solution
|
- For a homodiatomic molecule with a total number of electrons 14, after...
Text Solution
|
- The resonance hybrid of nitrate ion is
Text Solution
|
- Which one of the following is correct regarding sigma molecular orbita...
Text Solution
|
- pi(2px)^(**) differs from pi(2py)^(**) molecular orbital in which of t...
Text Solution
|
- The bond order of individual carbon bonds in benzene is
Text Solution
|
- In the formation of a homo diatomic neutral molecule, if N atomic orbi...
Text Solution
|
- In an anti bonding molecular orbital, electron density is minimum
Text Solution
|
- Which of the following pairs of species have the same bond order?
Text Solution
|
- The correct order of the energy of molecular orbitals in a molecules h...
Text Solution
|
- The molecular orbital electronic configuration is (sigma(1s))^(2),(sig...
Text Solution
|
- List -I " " List -II (Bonds) A. CO(2)" "1. 1, paramagnetic...
Text Solution
|