A N EXCEL PUBLICATION-MATRICES-QUESTION BANK
- Consider the following information regarding the purchase of pens and ...
Text Solution
|
- Construct a 2xx2 matrix A=[aij], whose (i,j)^th element is given by ...
Text Solution
|
- Given that a matrix A contains 15 elements what are the possible ord...
Text Solution
|
- Given that a matrix A contains 15 elements. Can A be a matrix with t...
Text Solution
|
- Construct a 3xx2 matrix whose elements are given by aij=1/2|i-3j|
Text Solution
|
- Find the values of a,b,c and d from the following equation: [[2a+b, ...
Text Solution
|
- In the matrix A=[[2,5,19,-7],[35,-2,5/2,12],[sqrt3,1,-5,17]] Write...
Text Solution
|
- In the matrix A=[[2,5,19,-7],[35,-2,5/2,12],[sqrt3,1,-5,17]] Write...
Text Solution
|
- In the matrix A=[[2,5,19,-7],[35,-2,5/2,12],[sqrt3,1,-5,17]] Write...
Text Solution
|
- If a matrix has 24 elements, What are the possible orders it can have?...
Text Solution
|
- If a matrix has 18 elements, What are the possible orders it can have?...
Text Solution
|
- Consider a 2xx2 matrix A=[a(ij)] , where A(ij)=(i+2j)^2/2 Write A
Text Solution
|
- Construct a 2xx2 matrix, A=[aij], whose elements are given by: aij=i...
Text Solution
|
- Consider a 2xx2 matrix A=[a(ij)] , where a(ij)=(i+2j)^2/2 Find A+A^T
Text Solution
|
- Find the value of x,y and z from the equation: [[4,3],[x,5]]=[[y,z],[1...
Text Solution
|
- Find the value of x,y and z from the equation: [[4,3],[x,5]]=[[y,z],[1...
Text Solution
|
- Find the value of x,y and z from the equation: [[x+y+z], [x+z],[y+z]]=...
Text Solution
|
- A=[aij](mxxn) is a square matrix, if a) m=n b) m>n c) m< n d) none o...
Text Solution
|
- The number of all possible 2xx2 matrices with entries 0 or 1 is
Text Solution
|
- Find x, y, z and w if [[x-y, 2x+z],[2x-y, 3z+w]]=[[-1,5],[0,13]]
Text Solution
|
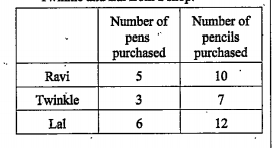 Represent the above data in the form of a `3xx2` matrix. What does the entry in the `2^nd` row and in the first column represent?
Represent the above data in the form of a `3xx2` matrix. What does the entry in the `2^nd` row and in the first column represent?