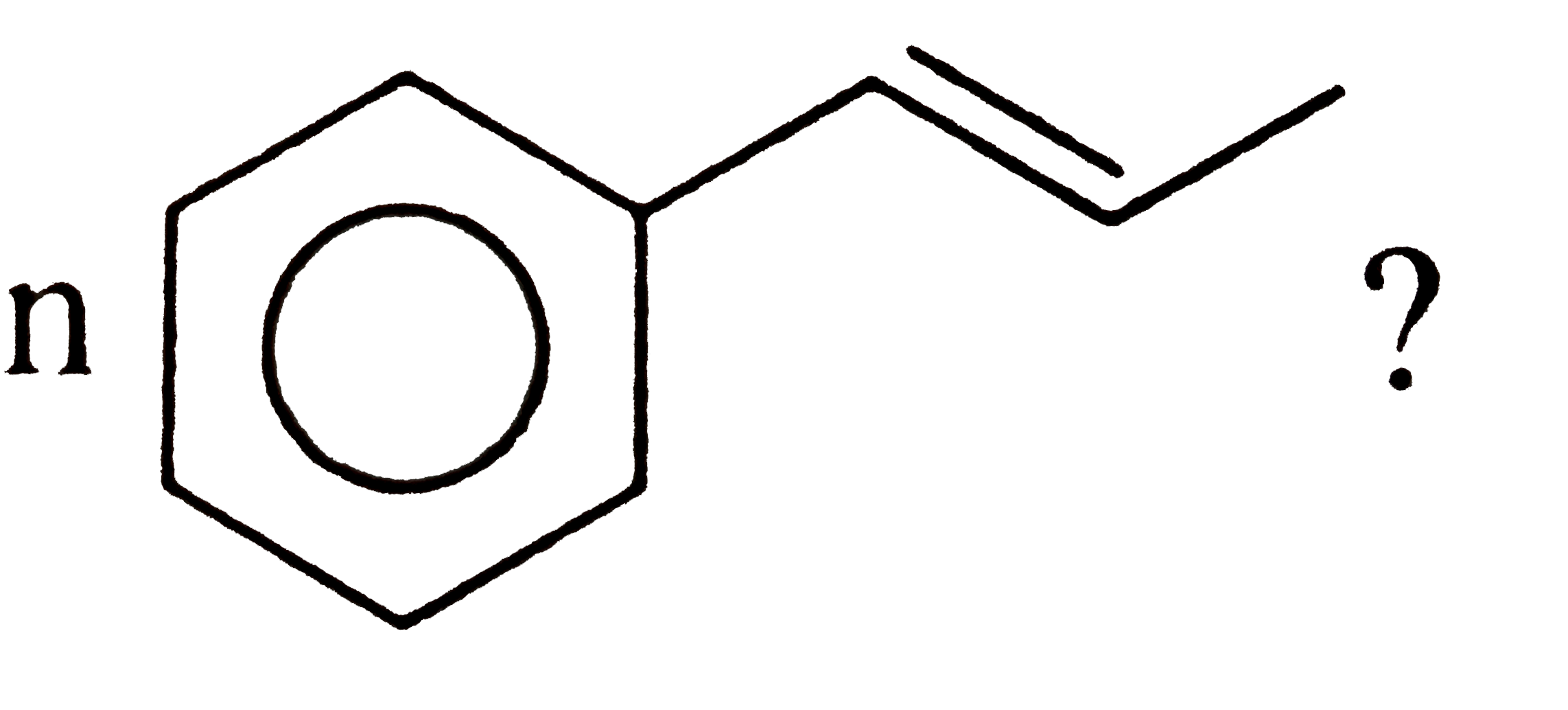
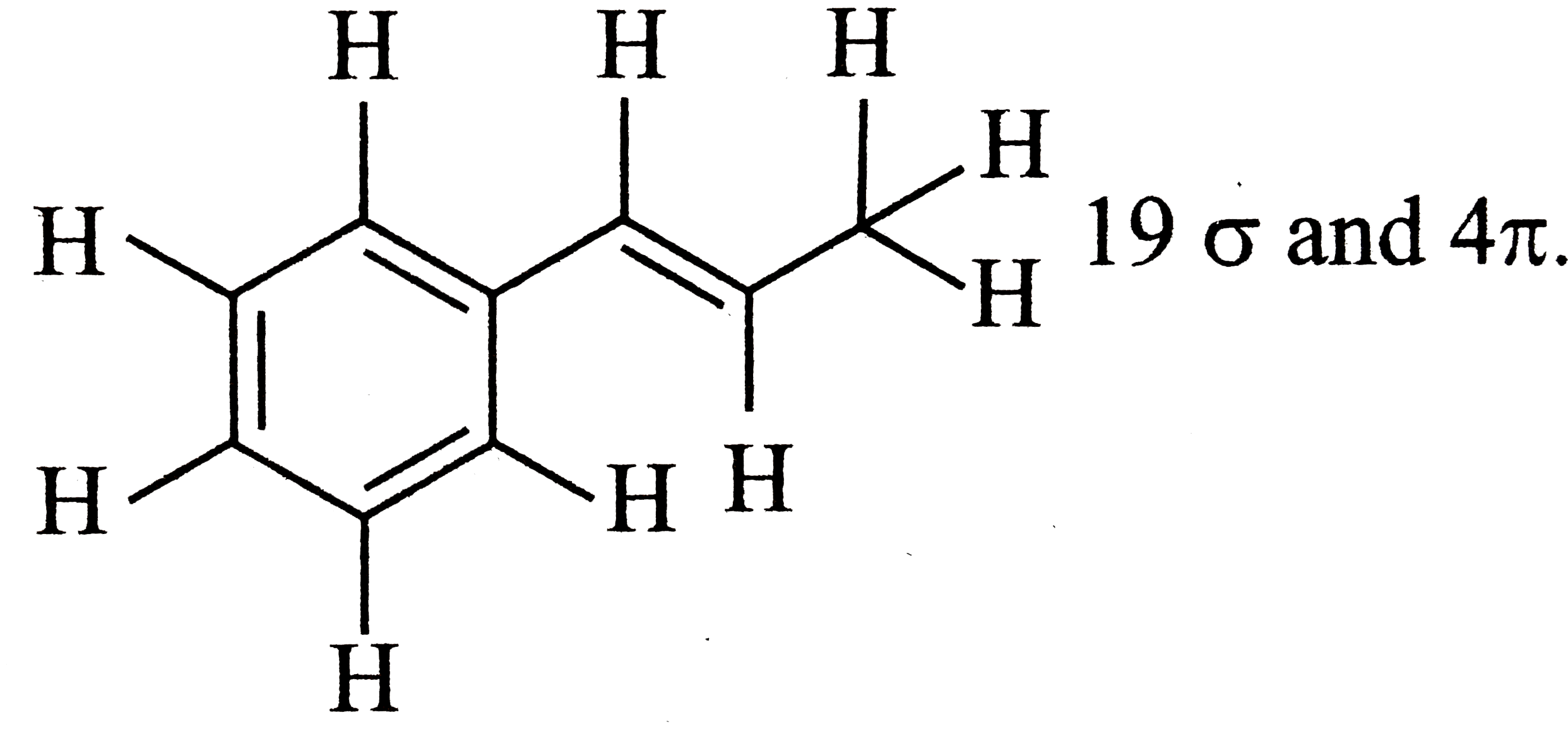
A
B
C
D
Text Solution
Verified by Experts
The correct Answer is:
Topper's Solved these Questions
CHEMICAL BONDING AND MOLECULAR STRUCTURE
A2Z|Exercise Vsepr Theory And Hybridisation|46 VideosCHEMICAL BONDING AND MOLECULAR STRUCTURE
A2Z|Exercise Molecular Orbitial Theory|42 VideosCHEMICAL BONDING AND MOLECULAR STRUCTURE
A2Z|Exercise Dipole Moment|28 VideosATOMIC STRUCTURE
A2Z|Exercise Section D - Chapter End Test|30 VideosCHEMICAL EQUILIBRIUM
A2Z|Exercise Section D - Chapter End Test|30 Videos
Similar Questions
Explore conceptually related problems
A2Z-CHEMICAL BONDING AND MOLECULAR STRUCTURE-Valence Bond Theory (Vbt)
- Which of the following are wrong?
Text Solution
|
- Which of the following overlaps gives a sigma bond with x as internucl...
Text Solution
|
- Which of the statement is correct about SO(2)?
Text Solution
|
- Which p-orbitals overlapping would give the strongest bond?
Text Solution
|
- Ratio of sigma and pi bonds is maximum in
Text Solution
|
- Which of the following is True stetement?
Text Solution
|
- HCN and HNC moleculas have equal number of
Text Solution
|
- Allyl cyanide has
Text Solution
|
- Mg(2)C(1) reacts with water forming propyne C(3)^(4-) has
Text Solution
|
- The strength of bonds by overlapping of atomic orbitals is in order
Text Solution
|
- Effective overlapping will be shown by:
Text Solution
|
- Main axis of diatometic molecule is z, molecular orbatals p(x) and p(y...
Text Solution
|
- Which plot best represents the potential energy ( E) of two hydrogen a...
Text Solution
|
- A: tetracynomethance, B: Carbon dioxide , C: Benzene, D: 1, 3butadiene...
Text Solution
|
- How many bonds are there in
Text Solution
|
- How many sigma- and pi - bond are there in salicycle acid?
Text Solution
|
- The ratio of sigma and pi bond in benzene is
Text Solution
|
- The enolic form of acetone contains:
Text Solution
|
- Which connot be expained by VBT?
Text Solution
|
- Which of the following leads to bonding?
Text Solution
|