NCERT EXEMPLAR-STRUCTURE OF ATOM-All Questions
- What is the experimental evidence in support of the diea that electron...
Text Solution
|
- Out of electron and proton which one will have, a higher velocity to p...
Text Solution
|
- A hypothetical electromagnetic wave is shown in figure. Find out the w...
Text Solution
|
- Chlorophyll present in green leaves of plants absorbs light at 4.620xx...
Text Solution
|
- What is the difference between the terms orbit and orbital?
Text Solution
|
- Table-tennis ball has mass 10g and s peed of 90m/s. if speed can be me...
Text Solution
|
- The effect of uncertainty principle is significant only for motion of ...
Text Solution
|
- Hydrogen atom has only one electron, So, mutual repulsion between elec...
Text Solution
|
- Match the following species with their corresponding ground state elec...
Text Solution
|
- Match the quantum numbers with the information provided by these.
Text Solution
|
- Match the following rules with their statements.
Text Solution
|
- Match species given in column I with the electronic configuration giv...
Text Solution
|
- Assertion(A): All isotopes of a given element show the same type of ch...
Text Solution
|
- Assertion(A) Black body is an ideal body that emits and absorbs radiat...
Text Solution
|
- Assertion (A) It is impossible to determine the exact position and exa...
Text Solution
|
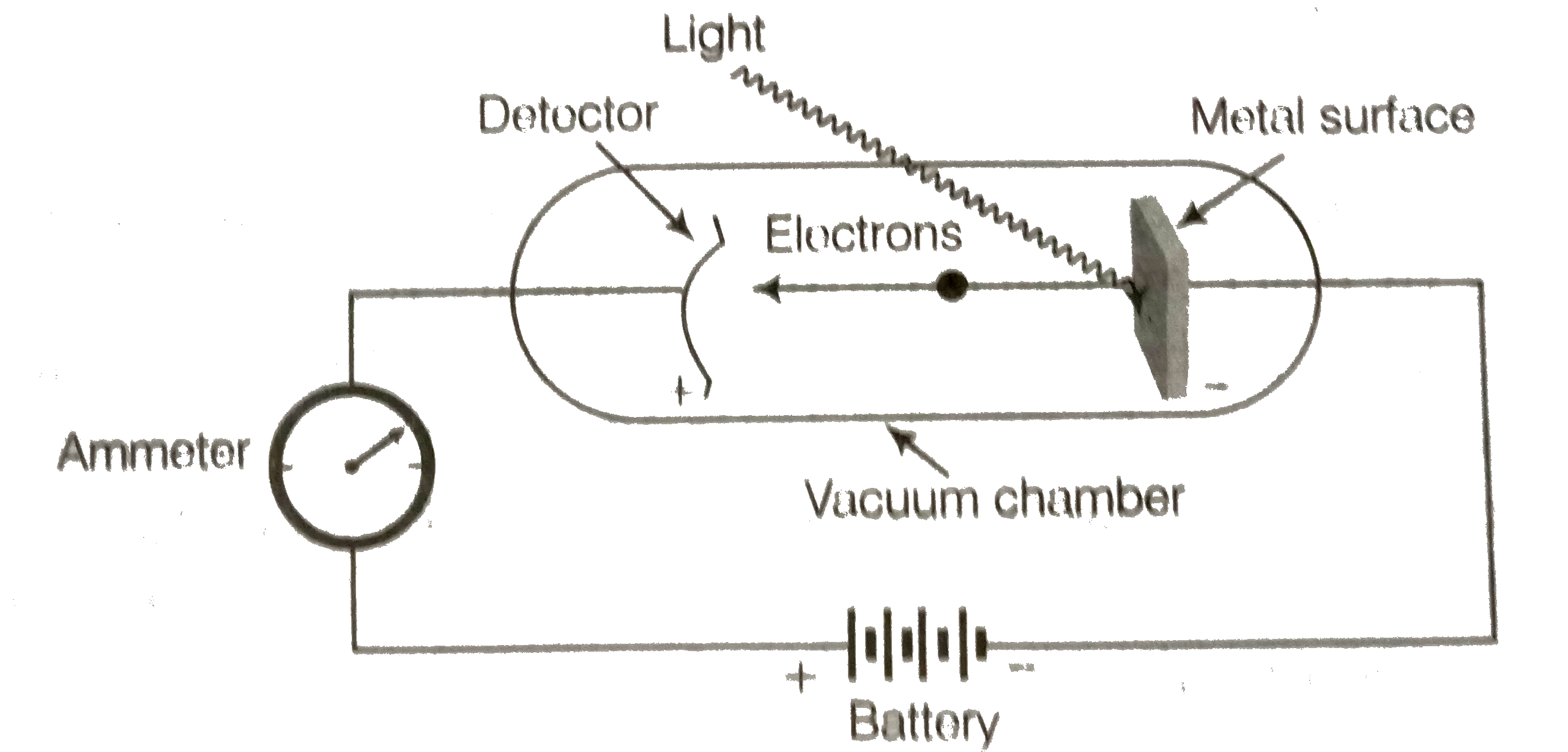
- What is photoelectric effect? State the result of photoelectric effect...
Text Solution
|
- Thershold frequency, v(0) is the minimum frequency which a photon must...
Text Solution
|
- When an electric discharge is passed through hydrogen gas, the hydroge...
Text Solution
|
- Calculate the energy and frequency of the radiation emitted when an el...
Text Solution
|
- Why was a chagne in the Bohr Model of atom required? Due to which impo...
Text Solution
|