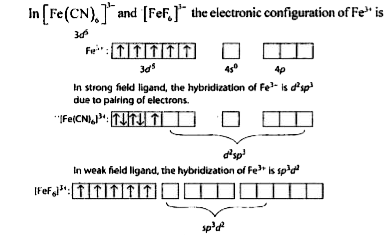
A
B
C
D
Text Solution
Verified by Experts
The correct Answer is:
Topper's Solved these Questions
COORDINATION COMPOUNDS
BRILLIANT PUBLICATION|Exercise LEVEL II|50 VideosCOORDINATION COMPOUNDS
BRILLIANT PUBLICATION|Exercise LEVEL III (Single Correct Answer Type )|10 VideosCOORDINATION COMPOUNDS
BRILLIANT PUBLICATION|Exercise LEVEL-II (ASSERTION- REASON TYPE )|20 VideosCO-ORDINATION COMPOUNDS AND ORGANOMETALLICS
BRILLIANT PUBLICATION|Exercise Level-II (Assertion- Reason)|4 VideosD & F BLOCK ELEMENTS
BRILLIANT PUBLICATION|Exercise Level -II|38 Videos
Similar Questions
Explore conceptually related problems
BRILLIANT PUBLICATION-COORDINATION COMPOUNDS -LEVEL I
- Sodium nitroprusside reacts with sulphide ion to give a purple colour ...
Text Solution
|
- Find the ligand having the highest denticity from the following option...
Text Solution
|
- Which of the following ligands is tridentate type?
Text Solution
|
- Which of the following complex has highest EAN value?
Text Solution
|
- Which of the following complexes contains a cationic ligand?
Text Solution
|
- In all the following complexes, the coordination number of iron is six...
Text Solution
|
- Consider the coordination compound, [Co(NH3)6]Cl3. In the formation of...
Text Solution
|
- The IUPAC name for the complex [Co(NO2)(NH3)5]Cl2 is
Text Solution
|
- Which among the following will be named as dibromidobis (ethylene diam...
Text Solution
|
- The IUPAC name of the complex [Ni (NH3)4][NiCl4]
Text Solution
|
- The correct statement about the magnetic properties of [Fe(CN)6]^(3-) ...
Text Solution
|
- Among the following species, the one which causes the highest CFSE, D...
Text Solution
|
- The ionization isomer of [Cr(H2O)4 Cl(NO2)]Cl is
Text Solution
|
- Choose the correct order for Delta@ for the following complexes. I) ...
Text Solution
|
- The IUPAC name of [Pt(NH3)4 (NO2)Cl] SO4 is
Text Solution
|
- Which of the following is not an example of sigma-bonded organometalli...
Text Solution
|
- Which of the folowing complex ions possesses sp^3 d^2 hybridization?
Text Solution
|
- The complex [Ni(dmg)2], where dmg is dimethylgloxime,
Text Solution
|
- Which of the following facts about the complex [Cr(NH3)6] Cl3 is wrong...
Text Solution
|
- The spectrochemical series of ligands is
Text Solution
|