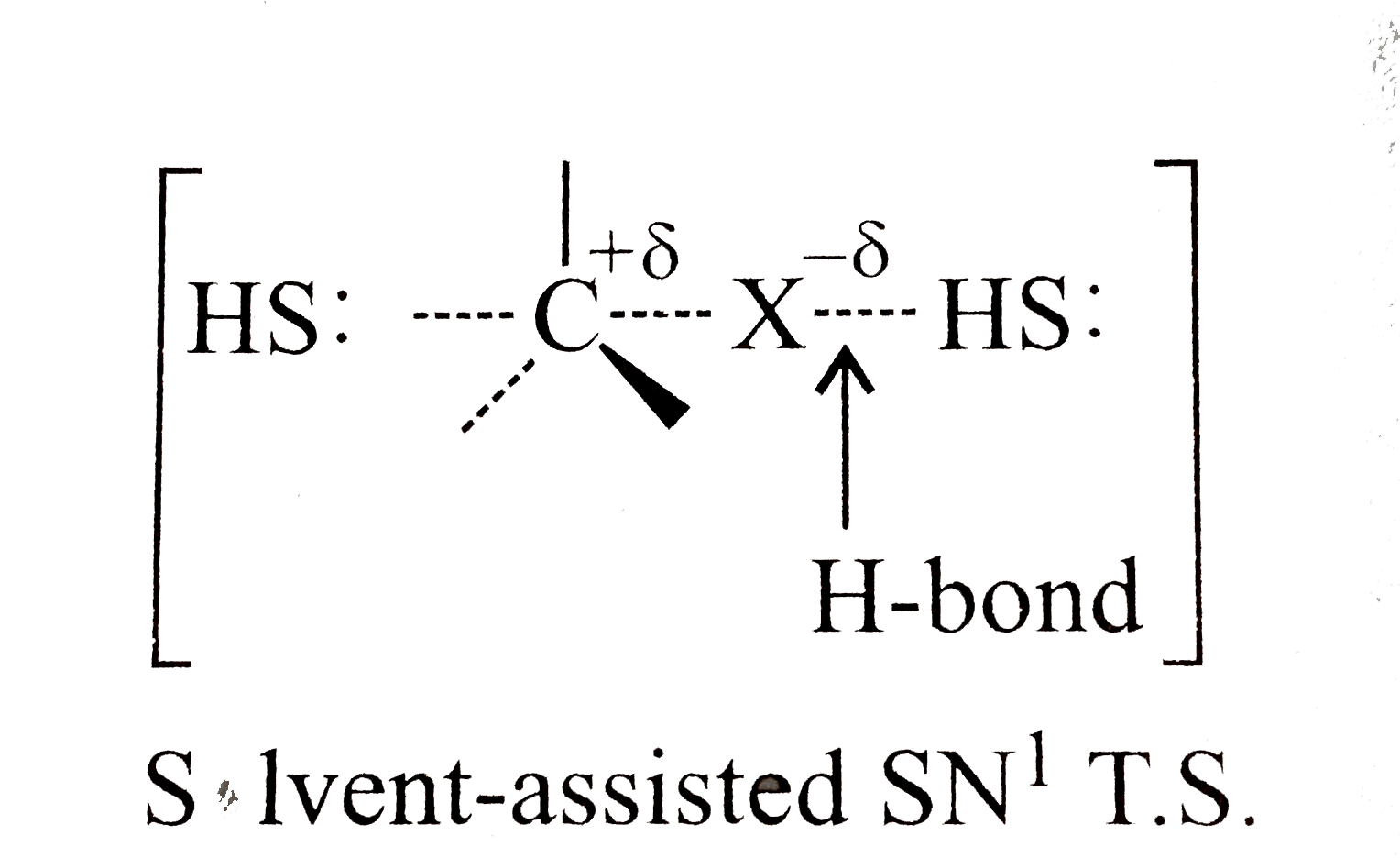Text Solution
Verified by Experts
Topper's Solved these Questions
ORGANIC REACTION MECHANISM
CENGAGE CHEMISTRY|Exercise Subjective|19 VideosORGANIC REACTION MECHANISM
CENGAGE CHEMISTRY|Exercise Comprehension|36 VideosORGANIC REACTION MECHANISM
CENGAGE CHEMISTRY|Exercise Analytical and Descriptive|6 VideosNCERT BASED EXERCISE
CENGAGE CHEMISTRY|Exercise Chemical Equilibrium|72 VideosP-BLOCK GROUP 13 - BORON FAMILY
CENGAGE CHEMISTRY|Exercise Exercise Archives (Subjecive)|9 Videos
Similar Questions
Explore conceptually related problems
CENGAGE CHEMISTRY-ORGANIC REACTION MECHANISM-Solved Example
- (a) Explain how the sterochemistry of SN^1 and SN^2 differs. (b) Exp...
Text Solution
|
- (a) Define dielectric constant. (b) How does dielectric constant aff...
Text Solution
|
- (a) Show by enthalpy diagram the rate of solvolysis of t-BuCl on incre...
Text Solution
|
- Hydrolysis of 2-bromo-3-methyl butane (2^@) yields only 2-methy-2-buta...
Text Solution
|
- Write structure for the solvolysis of Et3 C-Cl with : (a) MeOH (b)...
Text Solution
|
- Explain the relative rates of RX with H2 O/EtOH at 25^@ C as given : ...
Text Solution
|
- Predict the order of nucleophicity of X^(Ө) in the following reaction ...
Text Solution
|
- Give the product of the following displacement reactions : (a) (R )-...
Text Solution
|
- Explain .
Text Solution
|
- When 3-chlorocyclopropene (A) is treated with SbCl5, it gives a stable...
Text Solution
|
- Explain : (a) An aq. Solution to troplylium bormide (C7 H7Br) on tre...
Text Solution
|
- Give the order of reactivity towards aqueous HCOOH of the following. ...
Text Solution
|
- Give the order of hydrolysis in SN^1 process for the following : ...
Text Solution
|
- Give the order of (a) nucleophilicity (b) basic character, and (c) fug...
Text Solution
|
- Give the most reactive substrate in each of the following pair with ov...
Text Solution
|
- Outline the synthesis of following compounds from suitable nucleophile...
Text Solution
|
- Give a suitable mechanism for the following : .
Text Solution
|
- Predict the order of reactivity of the following halides with (a) NaI ...
Text Solution
|
- Explain : (a)ClCH2OCH3 (chloromethyl methyl ether) undergoes SN^1 re...
Text Solution
|
- Write the products of the following SN reactions. .
Text Solution
|