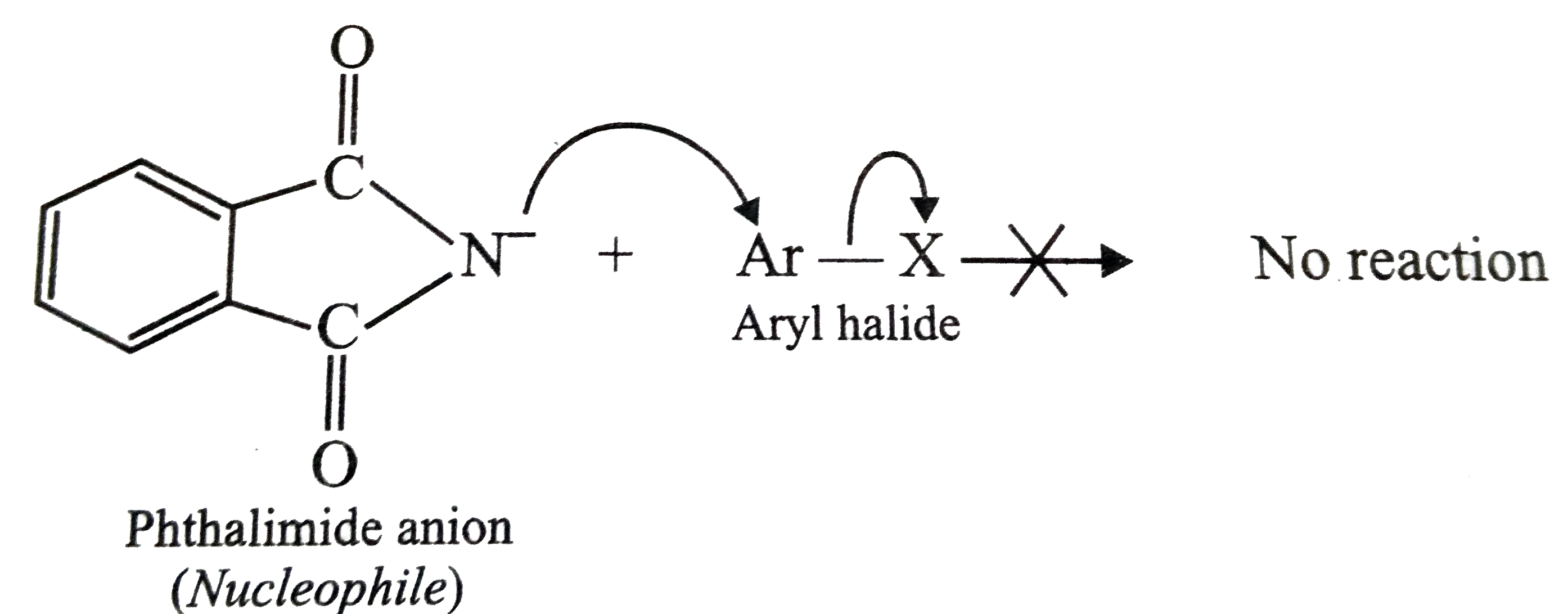Text Solution
Verified by Experts
Topper's Solved these Questions
ORGANIC COMPOUNDS CONTAINING NITROGEN
PRADEEP|Exercise NCERT EXEMPLANAR PROBLEMS WITH ANSWERS, HINTS AND SOLUTIONS (MUTIPLE CHOICE QUESTIONS-I)|27 VideosORGANIC COMPOUNDS CONTAINING NITROGEN
PRADEEP|Exercise NCERT EXEMPLANAR PROBLEMS WITH ANSWERS, HINTS AND SOLUTIONS (MUTIPLE CHOICE QUESTIONS-II)|10 VideosORGANIC COMPOUNDS CONTAINING NITROGEN
PRADEEP|Exercise NCERT QUESTIONS AND EXERCISES WITH ANSWERS (NCERT INTEXT UNSOLVED QUESTIONS)|9 VideosHALOALKANES AND HALOARENES
PRADEEP|Exercise IMPORTANT QUESTIONS FOR BOARD EXAMINATION|22 VideosP-BLOCK ELEMENTS
PRADEEP|Exercise IMPORTANT QUESTIONS FOR BOARD EXAMINATION|25 Videos
Similar Questions
Explore conceptually related problems
PRADEEP-ORGANIC COMPOUNDS CONTAINING NITROGEN-NCERT QUESTIONS AND EXERCISES WITH ANSWERS (NCERT EXERCISES)
- Write IUPAC names of the following compounds and classify them into pr...
Text Solution
|
- Give one chemical test to distinguish between the following pairs of c...
Text Solution
|
- Account for the following: (i) pK(b) of aniline is more than that of...
Text Solution
|
- Arrange the following: (i) in decreasing order of pK(a) values C(2)H...
Text Solution
|
- How will you convert (i) Ethanoic acid into methanamine. (ii) hexa...
Text Solution
|
- Describe a method for the identification of primary , secondary and te...
Text Solution
|
- Write short notes on the following : i. Carbylamine reaction ii....
Text Solution
|
- Accomplish the following conversions : i. Nitrobenzene to benzoic ac...
Text Solution
|
- If electronegativity of element A,B,C & D are 1.2, 3, 1.8 & 2.8 respec...
Text Solution
|
- An aromatic compound (A) on treatment with aqueous ammonia and heating...
Text Solution
|
- Complete the following reactions: (i) C(6)H(5)NH(2)+CHCl(3)+alc.KOHt...
Text Solution
|
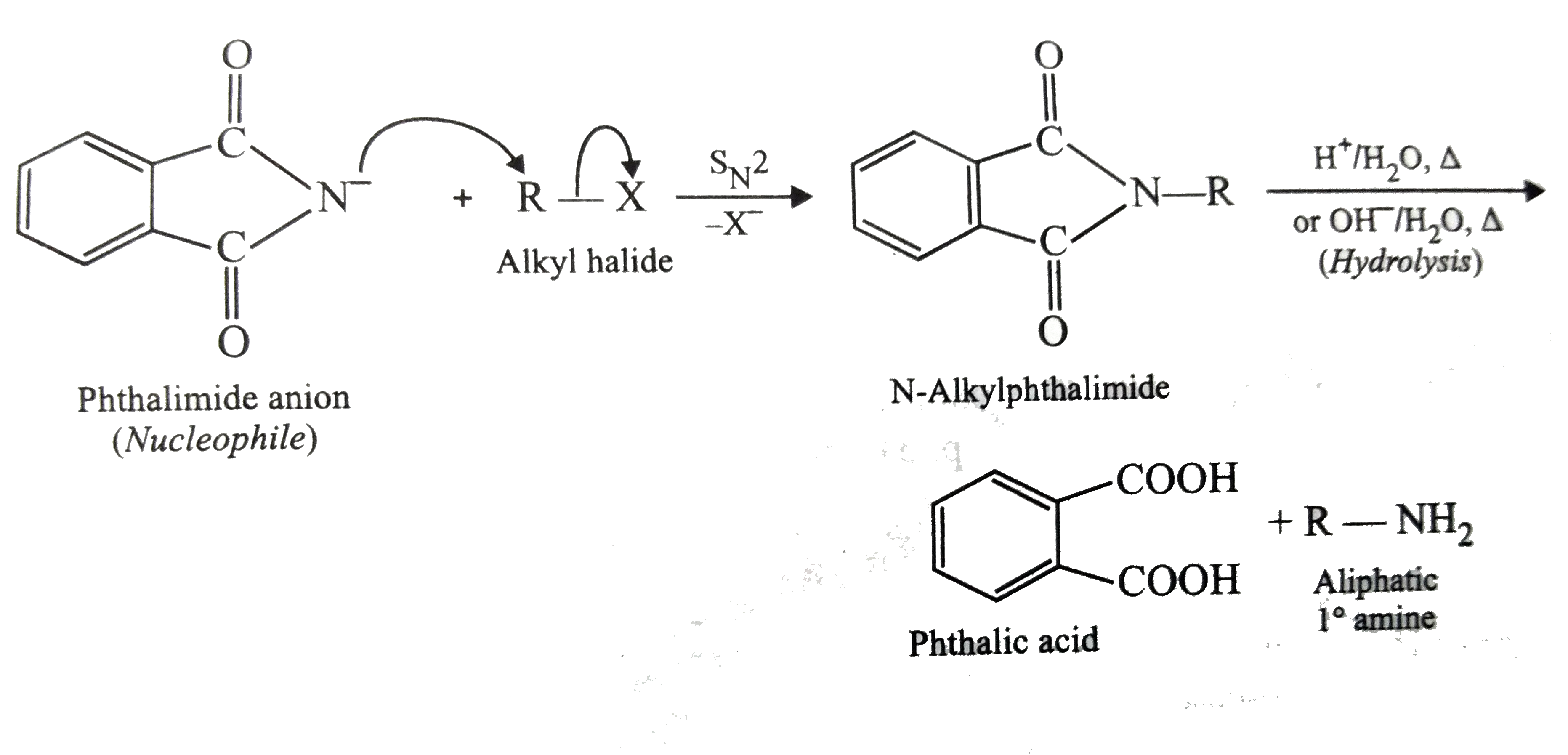
- Why cannot be aromatic primary amines prepared by Gabriel pthalimide s...
Text Solution
|
- Write the reaction of (i) aromatic and (ii) aliphatic primary amines...
Text Solution
|
- Give explanation for each of the following : (i) Why are amines les...
Text Solution
|