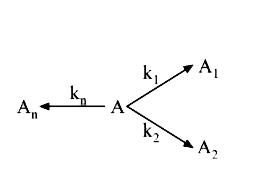
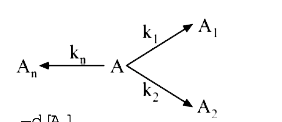
A
B
C
D
Text Solution
Verified by Experts
The correct Answer is:
Similar Questions
Explore conceptually related problems
Recommended Questions
- For a 1^(st) order decomposition, overall k will be given by :
Text Solution
|
- The correct order of 1^(st) ionisation enthalpy is :
Text Solution
|
- When rate = k[A]^(2)[B], the reaction is said to be of …………….order in ...
Text Solution
|
- For a Ist order decomposition of A as given Therefore rate constant (K...
Text Solution
|
- If the rate expression for a reaction is (dx)/(dt)=k[A]^(1//2)[B]^(2//...
Text Solution
|
- Foe the rate law, =K[A]^(3//2)[B]^(-1) the overall order of a reaction...
Text Solution
|
- The differential rate law of a reaction is given by, (dx)/(dt) =k[A]^1...
Text Solution
|
- Calculate the overall order of a reaction which has the rate expressio...
Text Solution
|
- Calculate the overall order of a reaction which has the rate expressio...
Text Solution
|