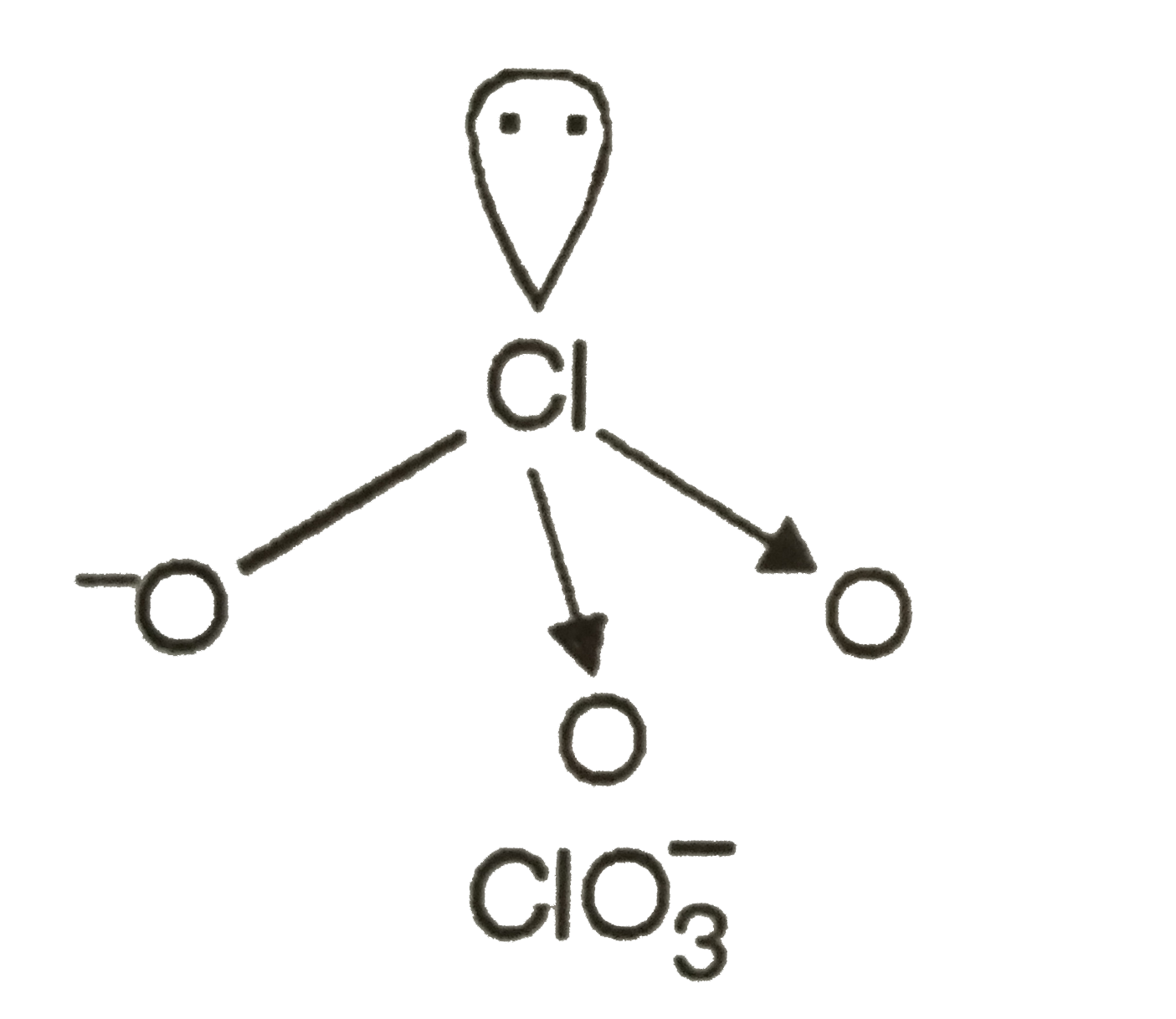
A
B
C
D
Text Solution
Verified by Experts
The correct Answer is:
Topper's Solved these Questions
THE HALOGEN FAMILY
DINESH PUBLICATION|Exercise Selected Straight Objective Type MCQs|37 VideosTHE HALOGEN FAMILY
DINESH PUBLICATION|Exercise MATRIX MATCH|2 VideosTHE HALOGEN FAMILY
DINESH PUBLICATION|Exercise ULTIMATE PREPARATORY PACKAGE|16 VideosTHE CARBON FAMILY
DINESH PUBLICATION|Exercise Ultimate|11 VideosTHE NITROGEN FAMILY
DINESH PUBLICATION|Exercise Ultimate|14 Videos
Similar Questions
Explore conceptually related problems
DINESH PUBLICATION-THE HALOGEN FAMILY-REVISION
- Which has maximum pH in aqueous solution ?
Text Solution
|
- Fluorine does not form oxyacids unlike other halogens because
Text Solution
|
- The shape of ClO3^- according to valence shell electron pair repulsion...
Text Solution
|
- The correct order of reactivity of halogens is
Text Solution
|
- Chlorine acts as a bleaching agent only in the presence of
Text Solution
|
- When chlorine reacts with cold and dilute solution of sodium hydroxide...
Text Solution
|
- Oxidation state of chlorine in hypochlorous acid
Text Solution
|
- In the reaction 2Br^(-) + X2 to Br2 + 2X^(-) , X2 is
Text Solution
|
- Chlorine is liberated, when we heat
Text Solution
|
- Which of the following halogens does not form its oxyacids ?
Text Solution
|
- Which of the following is not the characteristic of interhalogen compo...
Text Solution
|
- Which of the following is correct about the reaction ? 3NaClO overse...
Text Solution
|
- Which of the following is used in the preparation of chlorine?
Text Solution
|
- Which bond is most polar?
Text Solution
|
- On heating KClO(3) we get:
Text Solution
|
- The correct order of increasing oxidising power
Text Solution
|
- Freon is used as a
Text Solution
|
- Which of the following halogen does not exhibit positive oxidation sta...
Text Solution
|
- KI and CuSO4 solutions when mixed give
Text Solution
|
- Which of the following reaction involves oxidation and reduction ?
Text Solution
|