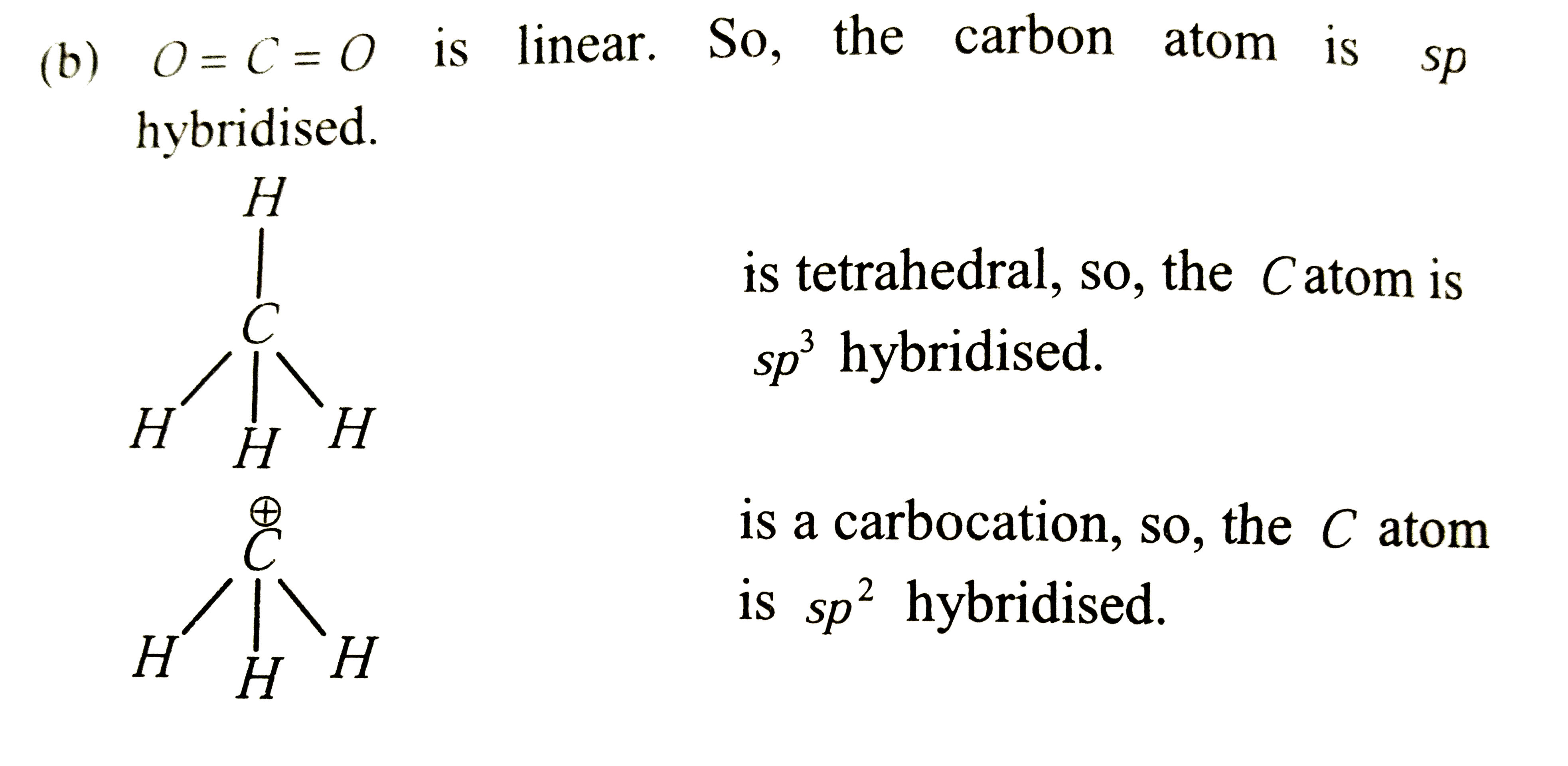A
B
C
D
Text Solution
Verified by Experts
The correct Answer is:
Similar Questions
Explore conceptually related problems
Recommended Questions
- In CO(2),CH(4) and CH(3)^(+) the hybridisation of carbon atoms are
Text Solution
|
- Hybridisation at 2nd carbon in CH(2)=CH-CH(3) is
Text Solution
|
- Mention the state of hybridisation of all the carbon atoms in he molec...
Text Solution
|
- Hybridisation of carbonium carbon in CH(3)^(+),CH(3)^(-)" and "CH(2)=C...
Text Solution
|
- In CO(2),CH(4) and CH(3)^(+) the hybridisation of carbon atoms are
Text Solution
|
- What are hybridisation states of each carbon atom in the following c...
Text Solution
|
- What is the type of hybridisation of each carbon in the following comp...
Text Solution
|
- CO(2), CH(4) तथा CH(3)^(+) में कार्बन का संकरण क्रमशः होगा :
Text Solution
|
- What are hybridisation states of each carbon atom in the following com...
Text Solution
|