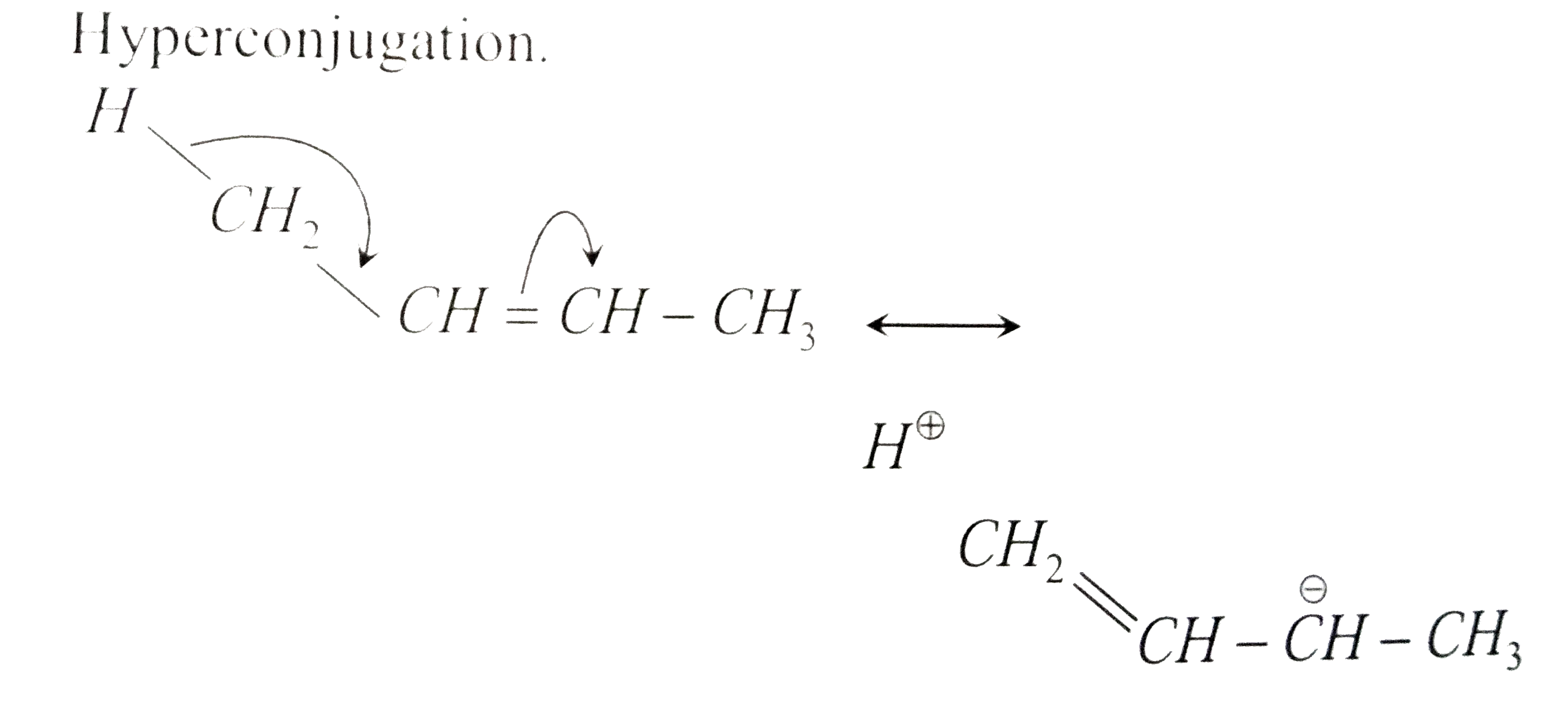
A
B
C
D
Text Solution
Verified by Experts
The correct Answer is:
Similar Questions
Explore conceptually related problems
Recommended Questions
- The hyperconjugative stabilities of tert-butyl cation and 2-butene, re...
Text Solution
|
- Among the given cations,……..is most stable (sec-bytyl carbonium ion, t...
Text Solution
|
- The hyperconjugative stabilities of tert-butyl cation and 2-butane, re...
Text Solution
|
- (A) Di-tert-butyl ether cannot be prepared by Williamson's ether synet...
Text Solution
|
- The hyperconjugative stabilities of tert-butyl cation and 2-butene, re...
Text Solution
|
- Statement-1. tert-Butyl carbanion is less stable than methyl carbanion...
Text Solution
|
- Tert-butyl chloride to 2-methylpropene
Text Solution
|
- In alkaline hydrolysis of tert-butyl bromide , the order of reaction w...
Text Solution
|
- The hyperconjugative stabilities of tert-butyl cation and 2-butene, re...
Text Solution
|