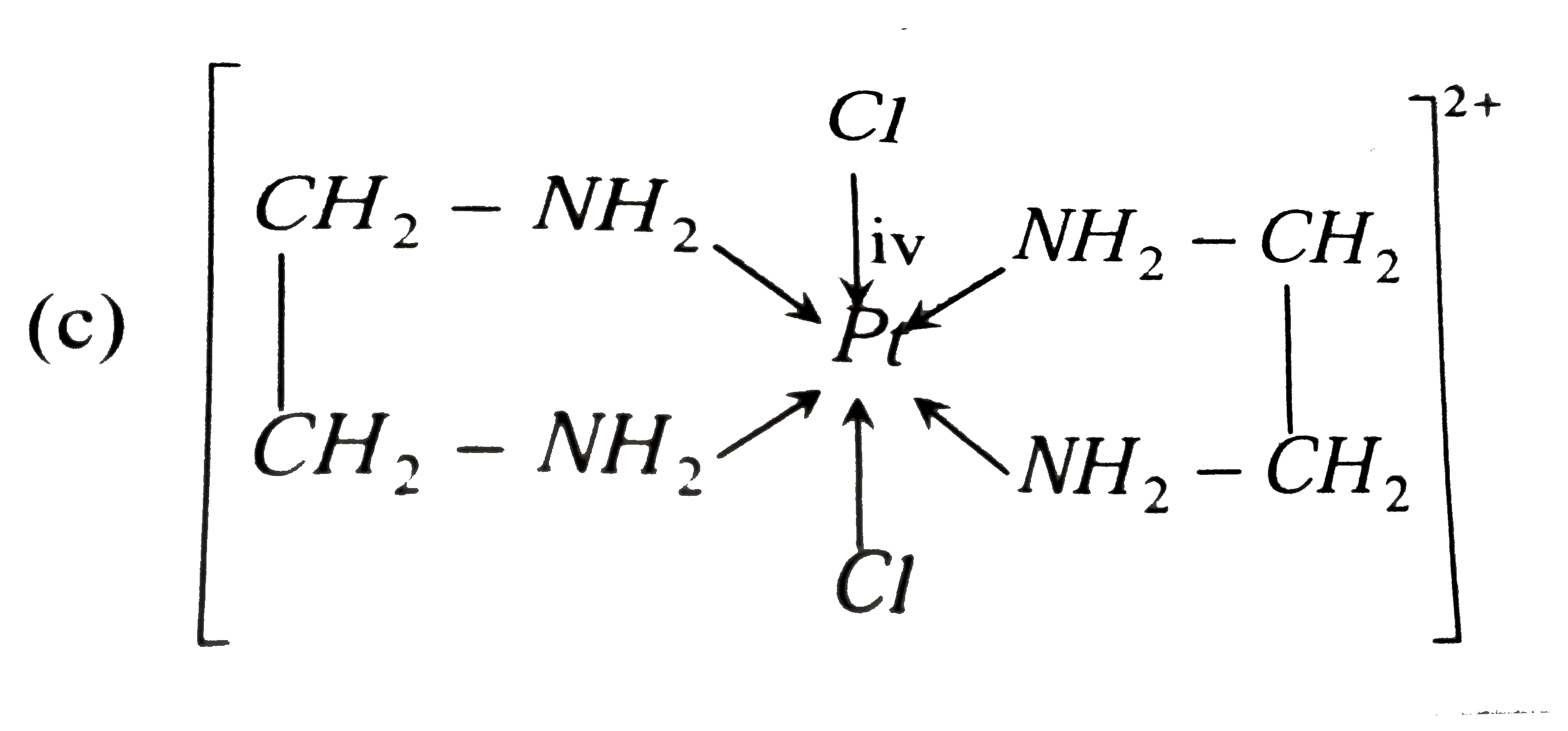
A
B
C
D
Text Solution
Verified by Experts
The correct Answer is:
Similar Questions
Explore conceptually related problems
Recommended Questions
- Double salts are addition compounds which lose their identity in aque...
Text Solution
|
- Choose the correct option for the complex [PtCl(2)(en)(2)]^(2+) .
Text Solution
|
- Double salts are addition compounds which lose their identity in aqueo...
Text Solution
|
- Double salts are addition compounds which lose their identity in aqueo...
Text Solution
|
- Double salts are addition compounds which lose their identity in aqueo...
Text Solution
|
- Double salts are addition compounds which lose their identity in aque...
Text Solution
|
- Double salts are addition compounds which lose their identity in aqueo...
Text Solution
|
- Double salts are addition compounds which lose their identify in aqueo...
Text Solution
|
- Double salts are addition compounds which lose their identify in aqueo...
Text Solution
|