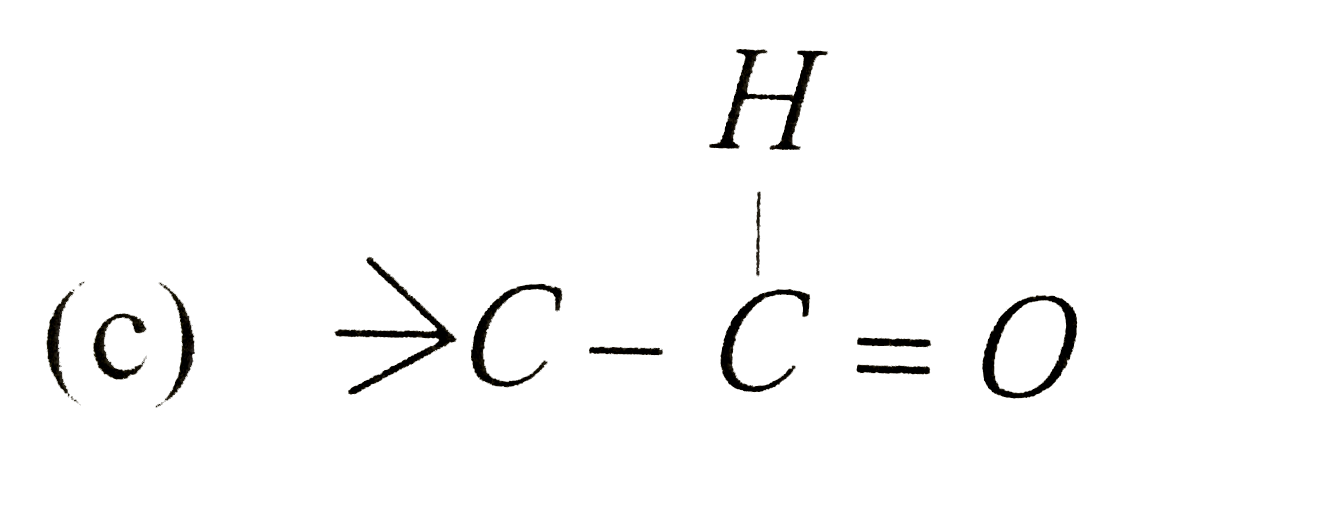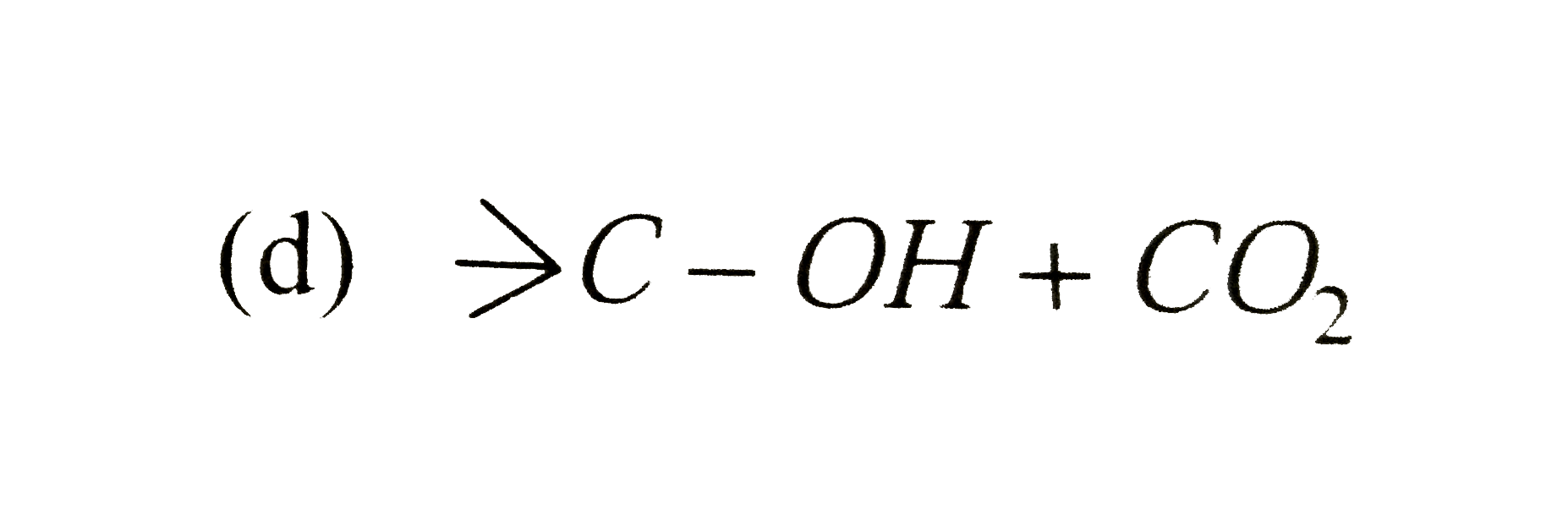A
B
C
D
Text Solution
AI Generated Solution
The correct Answer is:
Similar Questions
Explore conceptually related problems
Recommended Questions
- The ozonolysis of gt C= C lt produces
Text Solution
|
- Figure 2.23 shows the graph of the polynomial f(x)=a x^2+b x+c for wh...
Text Solution
|
- Write increasing order of basic strength of following: (i). (a) F^(ɵ) ...
Text Solution
|
- If a gt b, " then " (a)/(c) lt (b)/(c) for all a, b and c in R, where ...
Text Solution
|
- The ozonolysis of gt C= C lt produces
Text Solution
|
- The number of 7 digit integers abcdefg, where a lt b lt c lt d gt e gt...
Text Solution
|
- Which one of the following indicates the correct order of atomic size?...
Text Solution
|
- Which one of the following indicates the correct order of atomic size?...
Text Solution
|
- If a lt b and clt0, then (a)/(c)gt(b)/(c)
Text Solution
|