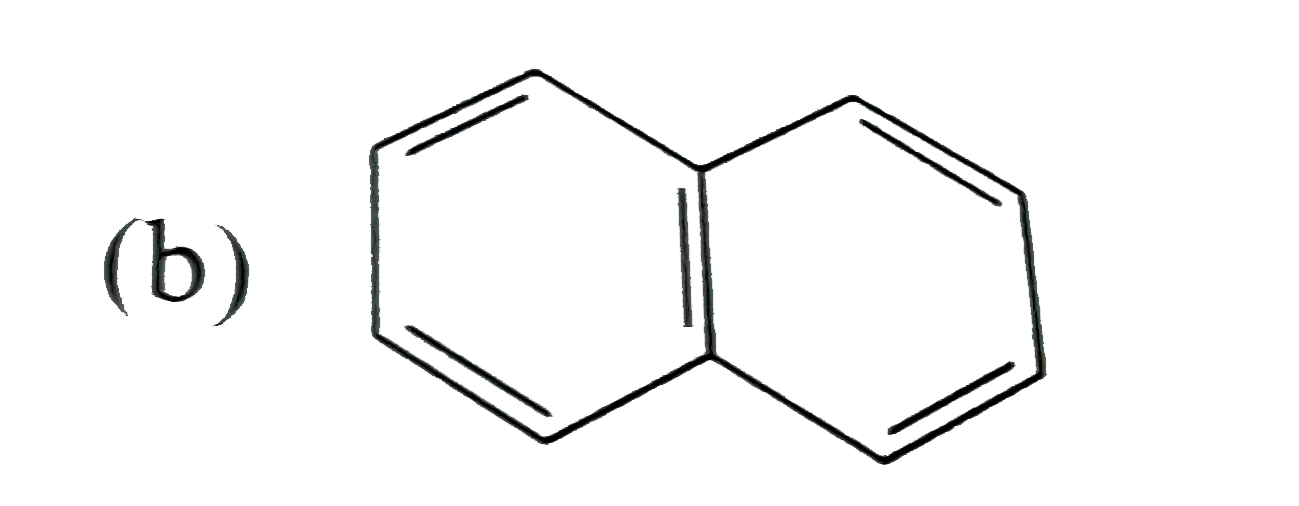
A
B
C
D
Text Solution
AI Generated Solution
The correct Answer is:
Similar Questions
Explore conceptually related problems
Recommended Questions
- The compound most likely to decolourize a solution of potassium perman...
Text Solution
|
- The compound most likely to decolourize a solution of potassium perman...
Text Solution
|
- An acidified solution of potassium permanganate oxidizes
Text Solution
|
- What is colour of Potassium permanganate solution
Text Solution
|
- 10 ml of a solution of H(2)O(2) labelled '10 volume' just decolourize...
Text Solution
|
- The alkaline potassium permanganate solution is known as .........
Text Solution
|
- On heating potassium permanganate, one of the following compound is no...
Text Solution
|
- एथिल बेन्जीन के पोटैशियम परमैंगनेट ऑक्सीकरण से यौगिक बनता है
Text Solution
|
- An acidified solution of potassium permanganate oxidizes
Text Solution
|