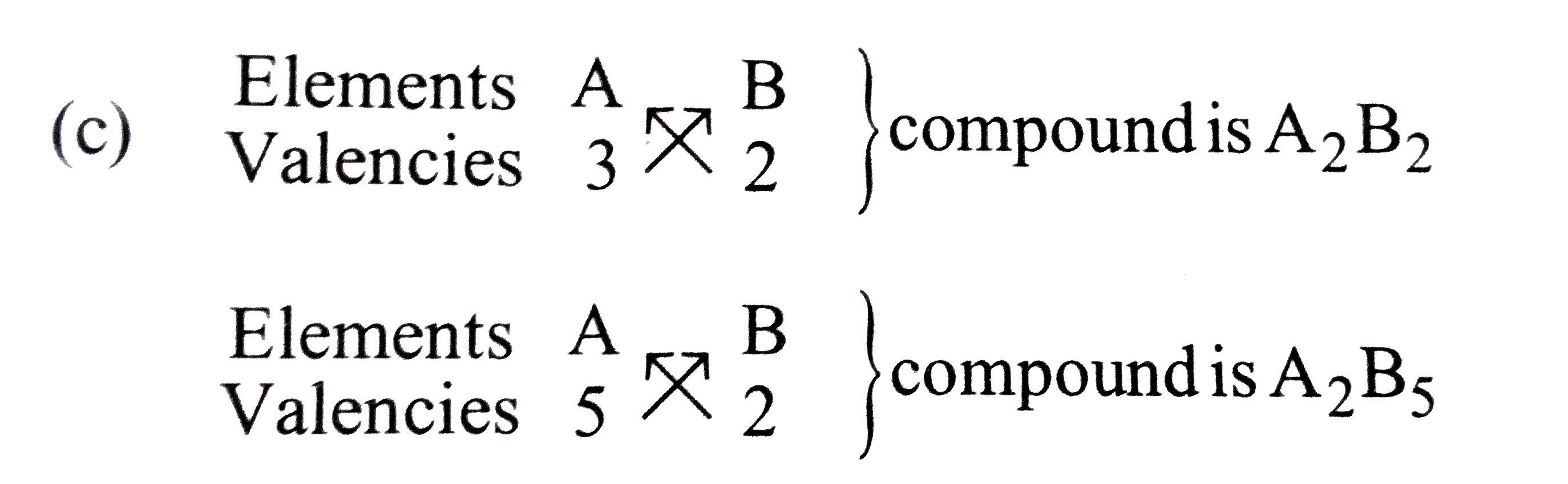A
B
C
D
Text Solution
Verified by Experts
The correct Answer is:
Similar Questions
Explore conceptually related problems
Recommended Questions
- An element A has valencies equal to 3 and 5. It combines with another ...
Text Solution
|
- The valency of an element X is 1 and that of oxygen is 2. What will b...
Text Solution
|
- An element X has a valency of 4 whereas another element Y has a valen...
Text Solution
|
- An element X of valency 3 conbines with another element Y of valenc...
Text Solution
|
- An element A forms an oxide A(2)O(5) . (a) What is the valency of elem...
Text Solution
|
- An element X has valency 3 while the element Y has valency 2. Write th...
Text Solution
|
- An element A has valencies equal to 3 and 5. It combines with another ...
Text Solution
|
- What would be the formula of the compound formed by A and B, where A h...
Text Solution
|
- तत्व A और B की संयोजकताएँ क्रमशः 3 और 2 हैं। इन तत्वों के संयोग से...
Text Solution
|