Similar Questions
Explore conceptually related problems
Recommended Questions
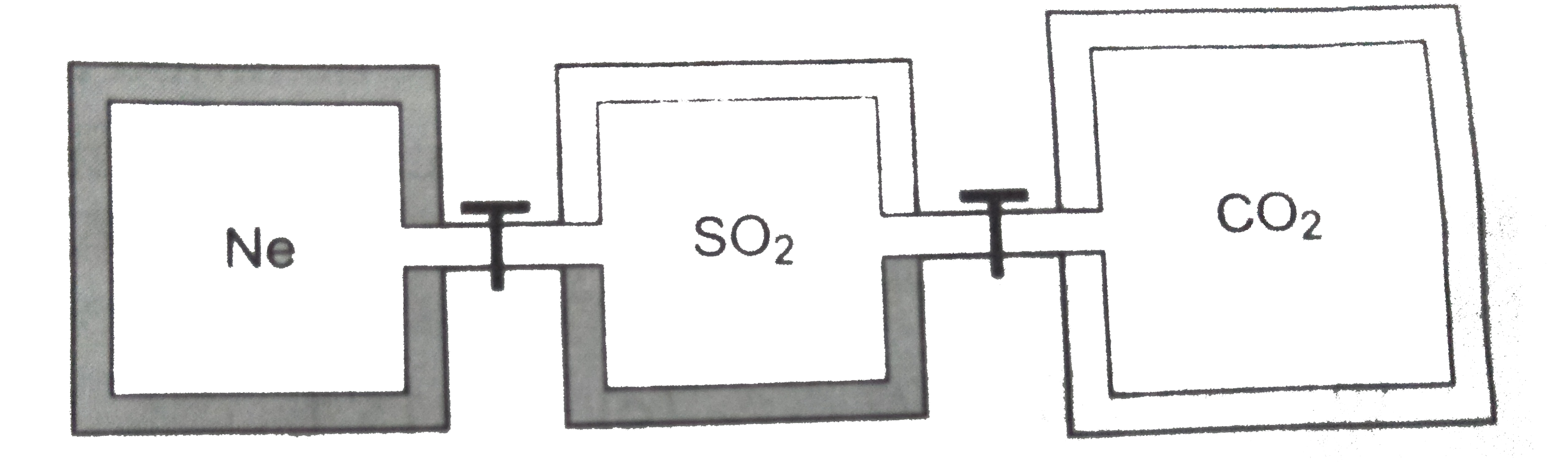
- Three closed rigid vessels, A, B and C, which initially contain three ...
Text Solution
|
- The volume of connecting tube is negligible. Now, the stopcock is open...
Text Solution
|
- Two rigid adiabatic vessel A and B which initially ,contain two gases ...
Text Solution
|
- Two closed vessel A and B of equal volume of 8.21 L are connected by a...
Text Solution
|
- Three closed rigid vessels, A, B and C, which initially contain three ...
Text Solution
|
- The temperature of a gas contain in a closed vessel increased by 2^(0)...
Text Solution
|
- A vessel of volume V contains a mixture of ideal gases at temperature ...
Text Solution
|
- A closed vessel contains 0.1 mole of a monatomic ideal gas at 200k . ...
Text Solution
|
- A vessel contains 1 mole of O2 at a temperature T. The pressure of gas...
Text Solution
|