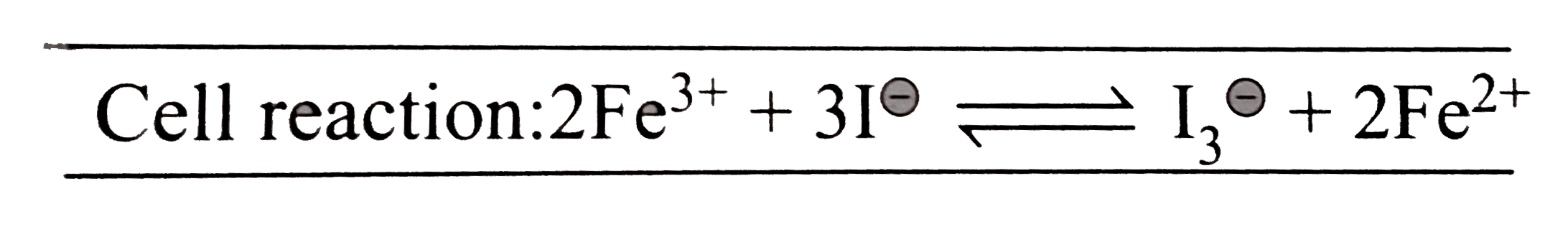Text Solution
Verified by Experts
The correct Answer is:
Topper's Solved these Questions
Similar Questions
Explore conceptually related problems
CENGAGE CHEMISTRY-ELECTROCHEMISTRY-Archieves Subjective
- A current of 1.70A is passed trhough 300.0 mL of 0.160M solution of Zn...
Text Solution
|
- For the galvanic cell: Ag|AgCI(s)|KCI(0.2M)||KBr(0.001M)|AgBr(s)|Ag, c...
Text Solution
|
- An aqueous solution of NaCl on electrolysis gives H(2)(g), Cl(2)(g), a...
Text Solution
|
- The standard reduction potential for the half cell : NO(3)^(-)(aq)+2...
Text Solution
|
- Chromium metal can be plated out from an acidic solution containing Cr...
Text Solution
|
- The standard reduction potential of the Ag^(o+)|Ag electrode at 298K i...
Text Solution
|
- The Edison storage cell is represented as : Fe(s)|FeO(s)|KOH(aq)|Ni(...
Text Solution
|
- An excess of liquid mercury is added to an acidicfied solution of 1.0x...
Text Solution
|
- The standard reduction potential for Cu^(2+)|Cu is +0.34V. Calculate t...
Text Solution
|
- How many grams of silver could be plated out on a serving tray by the ...
Text Solution
|
- Calculate the equilibrium constant for the reaction : Fe^(2+)+Ce^(4+)h...
Text Solution
|
- Calculate the equilibrium constant for the reaction : 2Fe^(3+)+3I^(Θ...
Text Solution
|
- Find the solubility product of a saturated solution of Ag(2)CrO(4) in ...
Text Solution
|
- A cell, Ag|Ag^(o+)||Cu^(2+)|Cu , initially contains 1 M Ag^(o+) and 1M...
Text Solution
|
- Copper sulphate solution (250 ML) was electrolyzed using a platinum an...
Text Solution
|
- The following electrochemical cell has been set up : Pt(l)|Fe^(3+),F...
Text Solution
|
- The standard potential of the following cell is 0.23 V at 15^(@)C and...
Text Solution
|
- Two students use same stock solution of ZnSO(4) and a solution of CuSO...
Text Solution
|
- Find the equilibrium constant for the reaction : Cu^(2+)+In^(2+)hArr...
Text Solution
|
- We have taken a saturated solution of AgBr, whose K(sp) is 12xx10^(-14...
Text Solution
|