Text Solution
Verified by Experts
Topper's Solved these Questions
COORDINATION COMPOUNDS
CENGAGE CHEMISTRY|Exercise Ex 7.1 Subjective (Terminology)|5 VideosCOORDINATION COMPOUNDS
CENGAGE CHEMISTRY|Exercise Ex 7.1 Subjective (Effective Atomic Number (Ean))|3 VideosCOORDINATION COMPOUNDS
CENGAGE CHEMISTRY|Exercise Archives Subjective|18 VideosCHEMICAL KINETICS
CENGAGE CHEMISTRY|Exercise Archives Subjective|23 VideosD AND F BLOCK ELEMENTS
CENGAGE CHEMISTRY|Exercise Archives Subjective|29 Videos
Similar Questions
Explore conceptually related problems
CENGAGE CHEMISTRY-COORDINATION COMPOUNDS-Solved Examples
- The enthalpy of hydration of the Fe^(2+) ion is 11.4kcal//mol higher t...
Text Solution
|
- If a complexing metal of the first transition series has a di configur...
Text Solution
|
- In terms of CFT explain why a d^(9) octahedral complex with six identi...
Text Solution
|
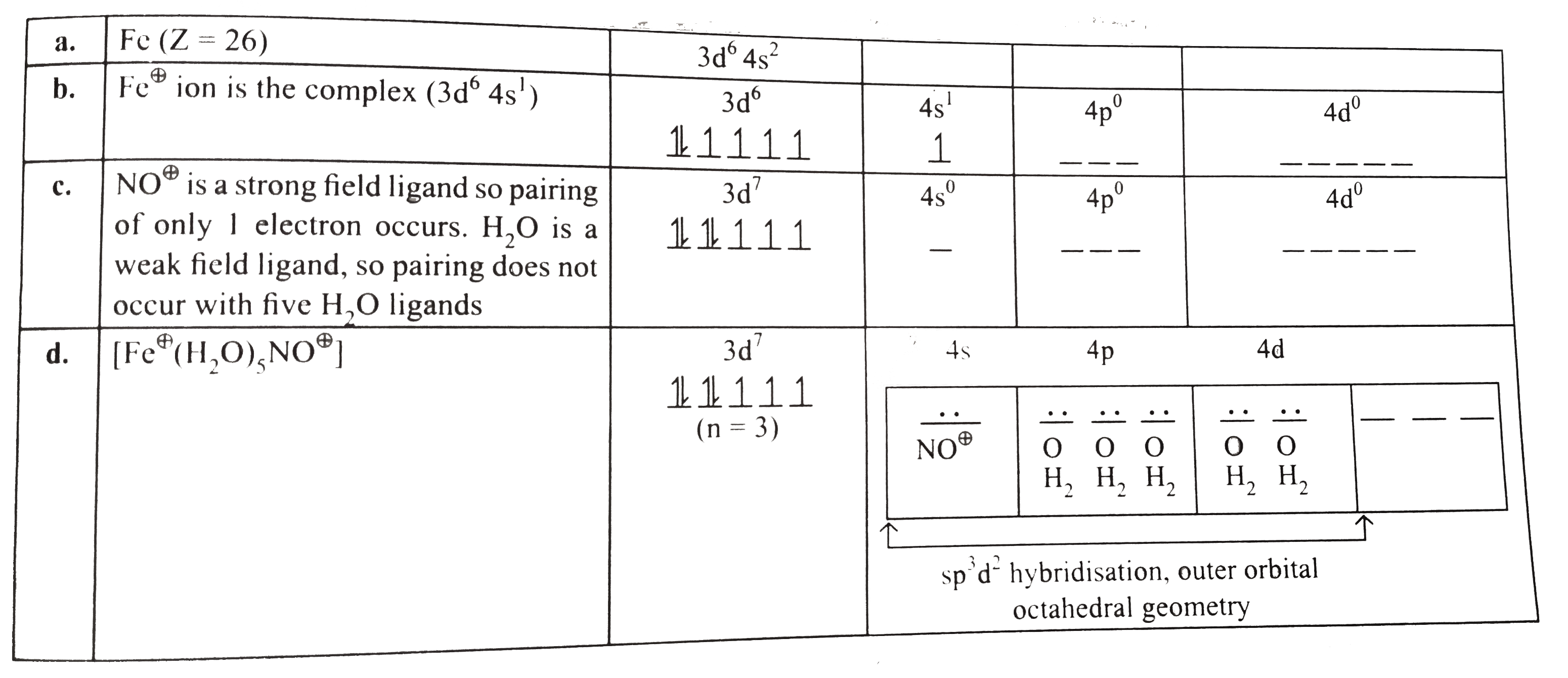
- Derive the geometry of the complex compound corresponding to the brown...
Text Solution
|
- Which of the electronic configuration according to crystal field theor...
Text Solution
|
- Which of the electronic configuration according to crystal field theor...
Text Solution
|
- According to crystal field therory the electronic configuration of the...
Text Solution
|
- According to crystal field theory the electronic configuration of the ...
Text Solution
|
- According to crystal field theory the electronic configuration of the ...
Text Solution
|
- Corystal field splitting energy (CFSE) for the complex [Cr(H(2)O)(6)]^...
Text Solution
|
- Crystal field splitting energy (CFSE) for the complex [Cr(NH(3))(6)]^(...
Text Solution
|
- Crystal field splitting energy (CFSE) for the complex [Cr(H(2)O)(6)]^(...
Text Solution
|
- Crystal field splitting energy (CFSE) for the complex [Cr(NH(3))(6)]^(...
Text Solution
|
- Crystal field splitting energey (CFSE) for the complex [Cr(NH(3)(6)]^(...
Text Solution
|
- In which structure crystal field splitting energey (CFSE) for octahedr...
Text Solution
|
- In which structure crystal field splitting energey (CFSE) for octahedr...
Text Solution
|
- Crystal field splitting energey (CFSE) for the complex [Fe(CN)(4)]^(Θ)...
Text Solution
|
- Crystal field splitting energey (CFSE) for the complex [Fe(O)(4)]^(2-)...
Text Solution
|
- CFSE for d^(6) octahedral complex having Delta =250 and P =125kJmo1^(-...
Text Solution
|
- The enthalpy of hydration of Cr^(+2) is -460 kcal mo1^(-1) In the abse...
Text Solution
|
 .
.