Text Solution
Verified by Experts
Topper's Solved these Questions
GENERAL ORGANIC CHEMISTRY
CENGAGE CHEMISTRY|Exercise Comprehension|20 VideosGENERAL ORGANIC CHEMISTRY
CENGAGE CHEMISTRY|Exercise Multiple Correct|22 VideosGENERAL ORGANIC CHEMISTRY
CENGAGE CHEMISTRY|Exercise Subjective|11 VideosCLASSIFICATION AND NOMENCLATURE OF ORGANIC COMPOUNDS
CENGAGE CHEMISTRY|Exercise Analytical and Descriptive Type|3 VideosHYDROGEN, WATER AND HYDROGEN PEROXIDE
CENGAGE CHEMISTRY|Exercise Subjective Archive (Subjective)|3 Videos
Similar Questions
Explore conceptually related problems
CENGAGE CHEMISTRY-GENERAL ORGANIC CHEMISTRY-Concept
- Using the curved-arrow notation, show the formation of reactive interm...
Text Solution
|
- Categorise the following molecules/ions ad nucleophile or electrophile...
Text Solution
|
- Identify electrophilic centre in the following : (1) CH3CH=O (2) C...
Text Solution
|
- Which bond is more polar in the following pairs of molecules ? (a) H...
Text Solution
|
- In which (C - C) bond of (H3 overset (3)C - overset (2) C H2 - overset...
Text Solution
|
- Write the resonance structures of (1) CH3 COO^(Ө) and (2) CH2 = CH - C...
Text Solution
|
- Explain why the following two structures (I) and (II) cannot be the ma...
Text Solution
|
- Explain why (CH3)3C^(oplus) is more stable than CH3 overset (oplus) C ...
Text Solution
|
- Draw the complete structures of bromomethane, bromethane, 2-bromopropa...
Text Solution
|
- Which is expected to be more stable, (I) O2NCH2CH2O^- or (II) CH3C...
Text Solution
|
- Draw the resonance structures for the following compounds. Show the el...
Text Solution
|
- Explain why alky1 groups act as electron donors when attacted to a pi-...
Text Solution
|
- Classify the reagents shown in bonds in the following equations as nuc...
Text Solution
|
- Classify the following in one of the reaction type studied. (a) CH3C...
Text Solution
|
- What is the relationship between the members of the following pairs of...
Text Solution
|
- For the following bond cleavages, use curved-arrow to show the electro...
Text Solution
|
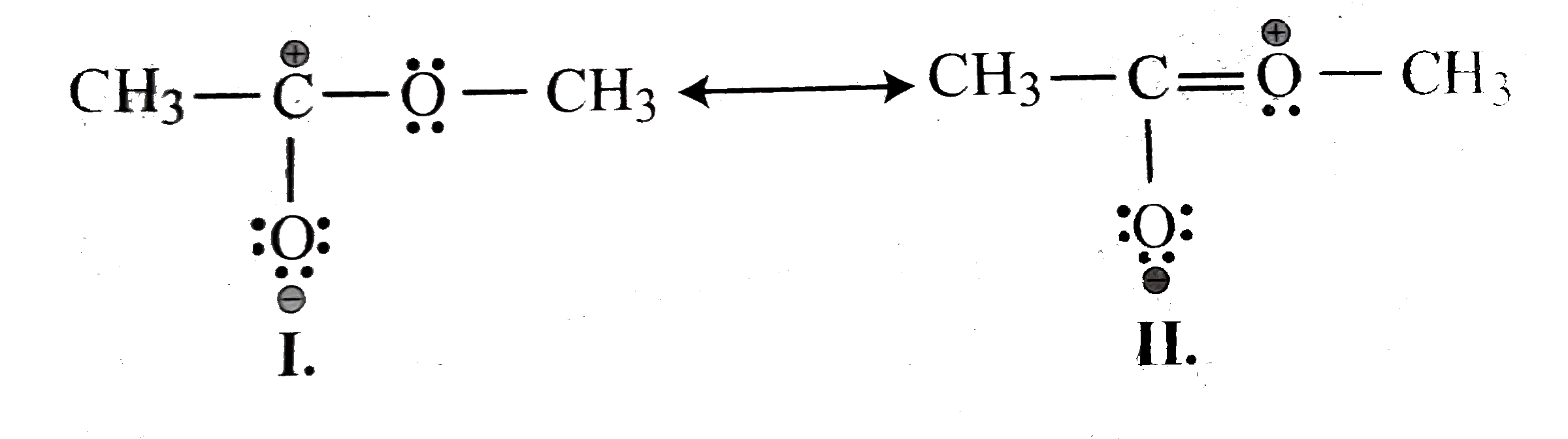 .
.