A
B
C
D
Text Solution
Verified by Experts
The correct Answer is:
Topper's Solved these Questions
CHEMICAL EQUILIBRIUM
A2Z|Exercise Degree Of Dissociation, Vapour Density And Simultaneous Equilibria|20 VideosCHEMICAL EQUILIBRIUM
A2Z|Exercise Section B - Assertion Reasoning|18 VideosCHEMICAL EQUILIBRIUM
A2Z|Exercise Application Of Equllibrium Constant (K)|65 VideosCHEMICAL BONDING AND MOLECULAR STRUCTURE
A2Z|Exercise Section D - Chapter End Test|30 VideosCHEMICAL THERMODYNAMICS
A2Z|Exercise Section D - Chapter End Test|30 Videos
Similar Questions
Explore conceptually related problems
A2Z-CHEMICAL EQUILIBRIUM-Le - Chatellers'S Principle
- Consider the reaction HCN((aq))hArrH((aq))^(+) + CN((aq))^(-) . At equ...
Text Solution
|
- Addition of water to which of the following equilibria causes it to s...
Text Solution
|
- In the gaseous equilibrium H(2)X(2) + heat hArr 2HX, the formation of ...
Text Solution
|
- The equilibrium constants for the reaction Br(2)hArr 2Br at 500 K an...
Text Solution
|
- Reasing the temperature of an equilibrium system
Text Solution
|
- Some quantity of water is contained in a container as shown in figure....
Text Solution
|
- When a bottle of cold drink is opened, the gas comes out with a fizzle...
Text Solution
|
- Reaction in which yield of product will increase with increase in pres...
Text Solution
|
- In the reaction A(2)(g) + 4B(2)(g)hArr2AB(4)(g), Delta H lt 0 . The de...
Text Solution
|
- The endothermic reaction (M+NhArrP)is allowed to attain an equilibrium...
Text Solution
|
- The equilibrium constant for a reacton N(2)(g)+O(2)(g)=2NO(g) is 4xx...
Text Solution
|
- According to Le-Chatelier's principle, an increase in the temperature ...
Text Solution
|
- In which of the following equilibrium, change in the volume of the sys...
Text Solution
|
- In which of the following reactions, increase in the pressure at tempe...
Text Solution
|
- Suppose the reaction PC1(5(s))hArrPC1(3(s)) + C1(2(g)) is in a closed ...
Text Solution
|
- Consider the reaction equilibrium, 2SO(2(g)) + O(2(g))hArr , Delta H^(...
Text Solution
|
- In which of the following equilibrium reactions, the equilibrium react...
Text Solution
|
- The exothermic formation of C1F(3) is represented by the equation C1(2...
Text Solution
|
- In which of the following gaseous equilibrium an increase in pressure ...
Text Solution
|
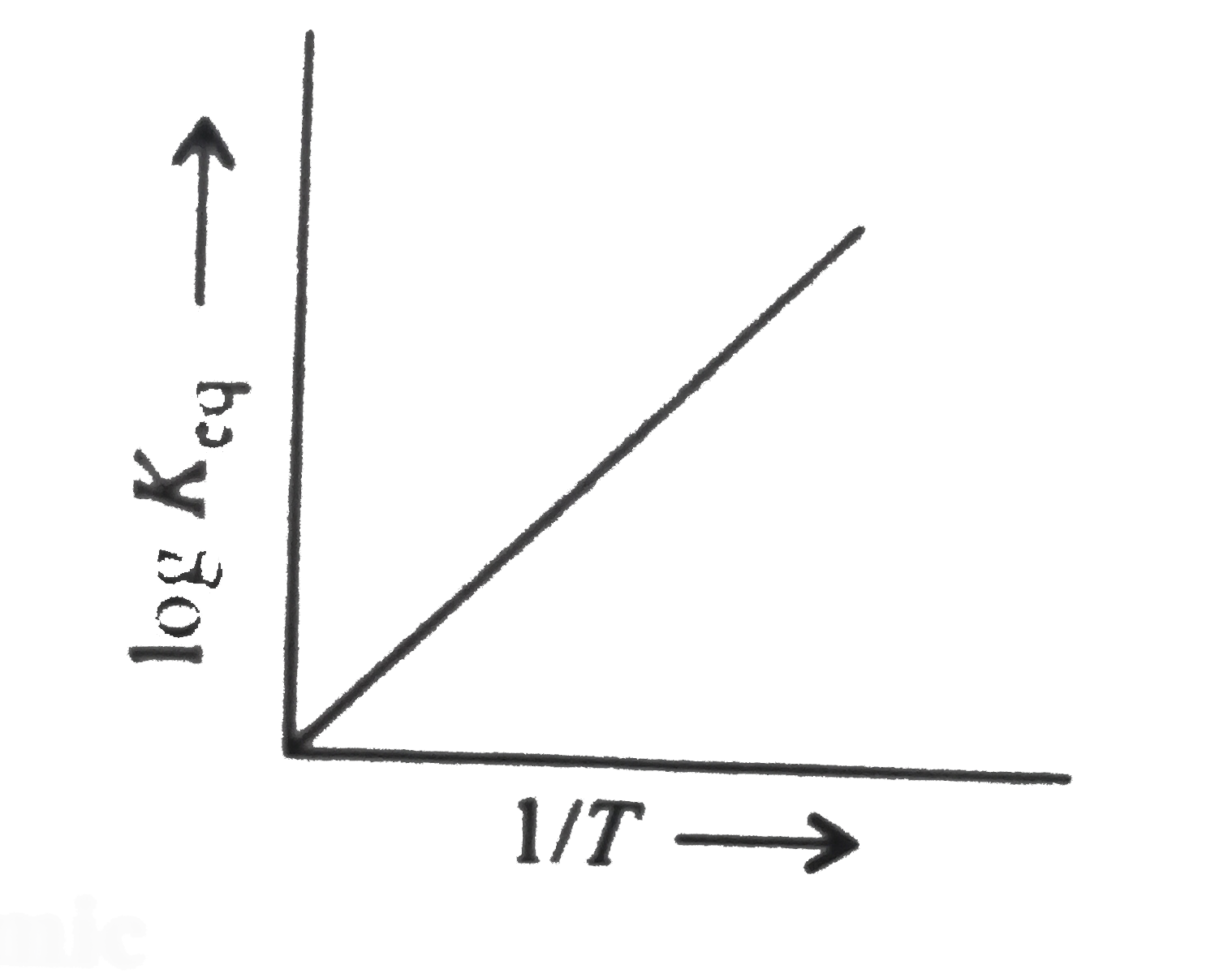
- A schematic plot of log K(eq) vs inverse of temperature for a reaction...
Text Solution
|