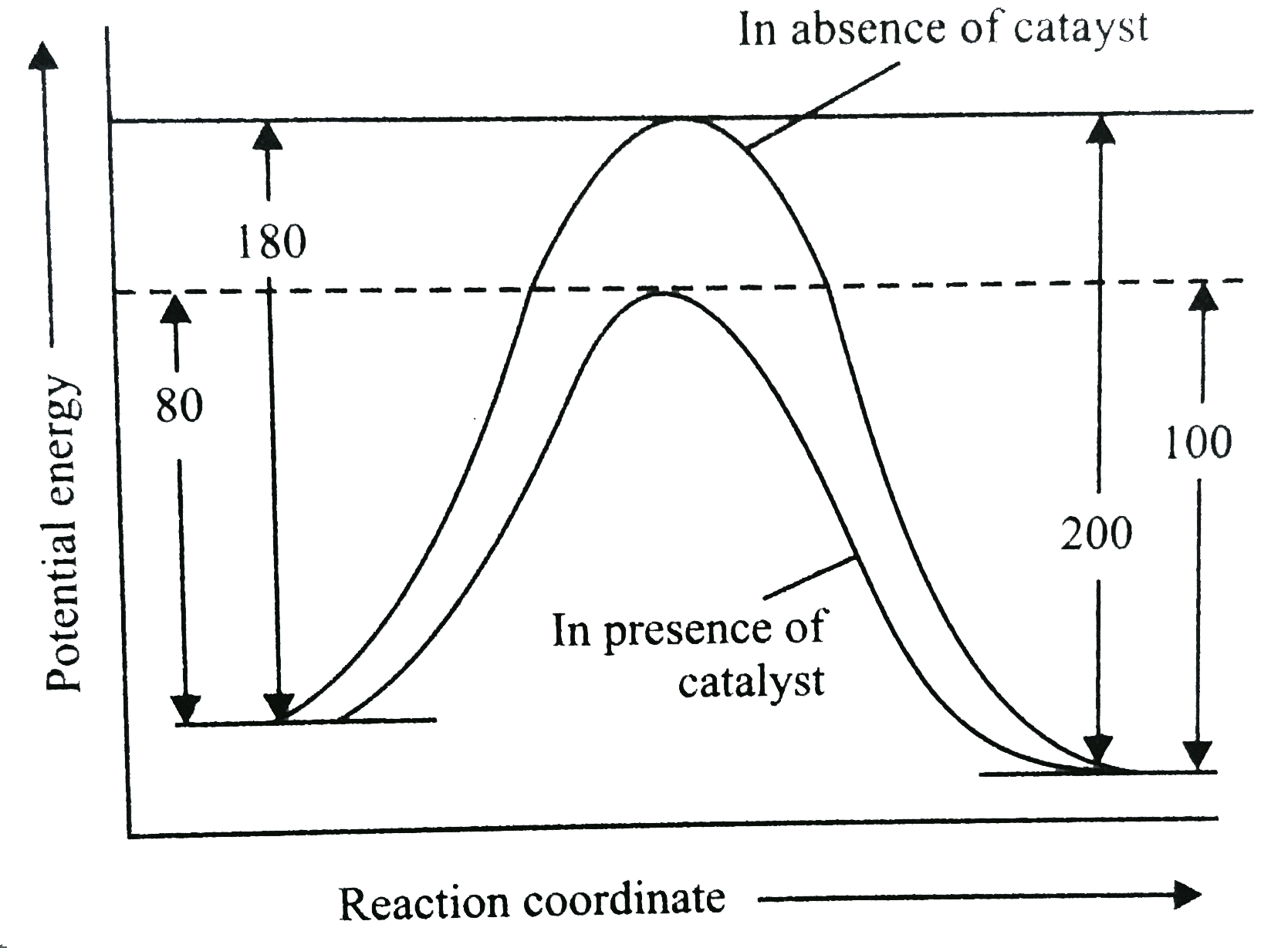
A
B
C
D
Text Solution
Verified by Experts
The correct Answer is:
Topper's Solved these Questions
CHEMICAL KINETICS
A2Z|Exercise Parallel Reaction, Consecutive Reaction, Special Cases Of First-Order Reactions|25 VideosCHEMICAL KINETICS
A2Z|Exercise Section B - Assertion Reasoning|21 VideosCHEMICAL KINETICS
A2Z|Exercise Initial Rate Method And Ostwaid Method|18 VideosBIOMOLECULES
A2Z|Exercise Section D - Chapter End Test|30 VideosCOORDINATION COMPOUNDS
A2Z|Exercise Section D - Chapter End Test|30 Videos
Similar Questions
Explore conceptually related problems
A2Z-CHEMICAL KINETICS-Arrhenius Equation, Effect Of Temprature And Effect Of Catalysts
- The Activation energy for a chemical reaction mainly depends upon
Text Solution
|
- A molecule of gas is struck by another molecule of the same gas, the f...
Text Solution
|
- Activation energy of a chemical reaction can be determined by
Text Solution
|
- The energies of activation for forward and reverse reaction for A(2)+...
Text Solution
|
- ArarrB, DeltaH= -10 KJ mol^(-1), E(a(f))=50 KJ mol^(-1), then E(a) of ...
Text Solution
|
- The rate of a reaction doubles when its temperature changes form 300 K...
Text Solution
|
- The rate of a chemical reaction doubles for every 10^(@)C rise of temp...
Text Solution
|
- A reactant (A) forms two products A overset (k(1))rarr B, Activation...
Text Solution
|
- The first-order rate constant k is related to temperature as log k = 1...
Text Solution
|
- The Arrhenius relationship of two different reactions is shown below. ...
Text Solution
|
- The rate constant of most of the reaction increases with increase in t...
Text Solution
|
- For a reaction, the rate constant is expressed as k = Ae^(-40000//T). ...
Text Solution
|
- The ratio of rate constant at 27^(@)C and 37^(@)C is Q(10). What shoul...
Text Solution
|
- The decomposition of N(2)O into N(2) & O(2) in presence of gaseous arg...
Text Solution
|
- Given that the temperature coefficient for the saponification of ethyl...
Text Solution
|
- A reaction takes place in three steps: the rate constant are k(1), k(2...
Text Solution
|
- How much faster would a reaction proceed at 25^(@)C than at 0^(@)C if ...
Text Solution
|
- By increasing the temperature by 10^(@)C, the rate of forward reaction...
Text Solution
|
- The distribution of molecular kinetic energy at two temperature is as ...
Text Solution
|
- The rate of a reaction gets doubled when the temperature changes from ...
Text Solution
|