Similar Questions
Explore conceptually related problems
Recommended Questions
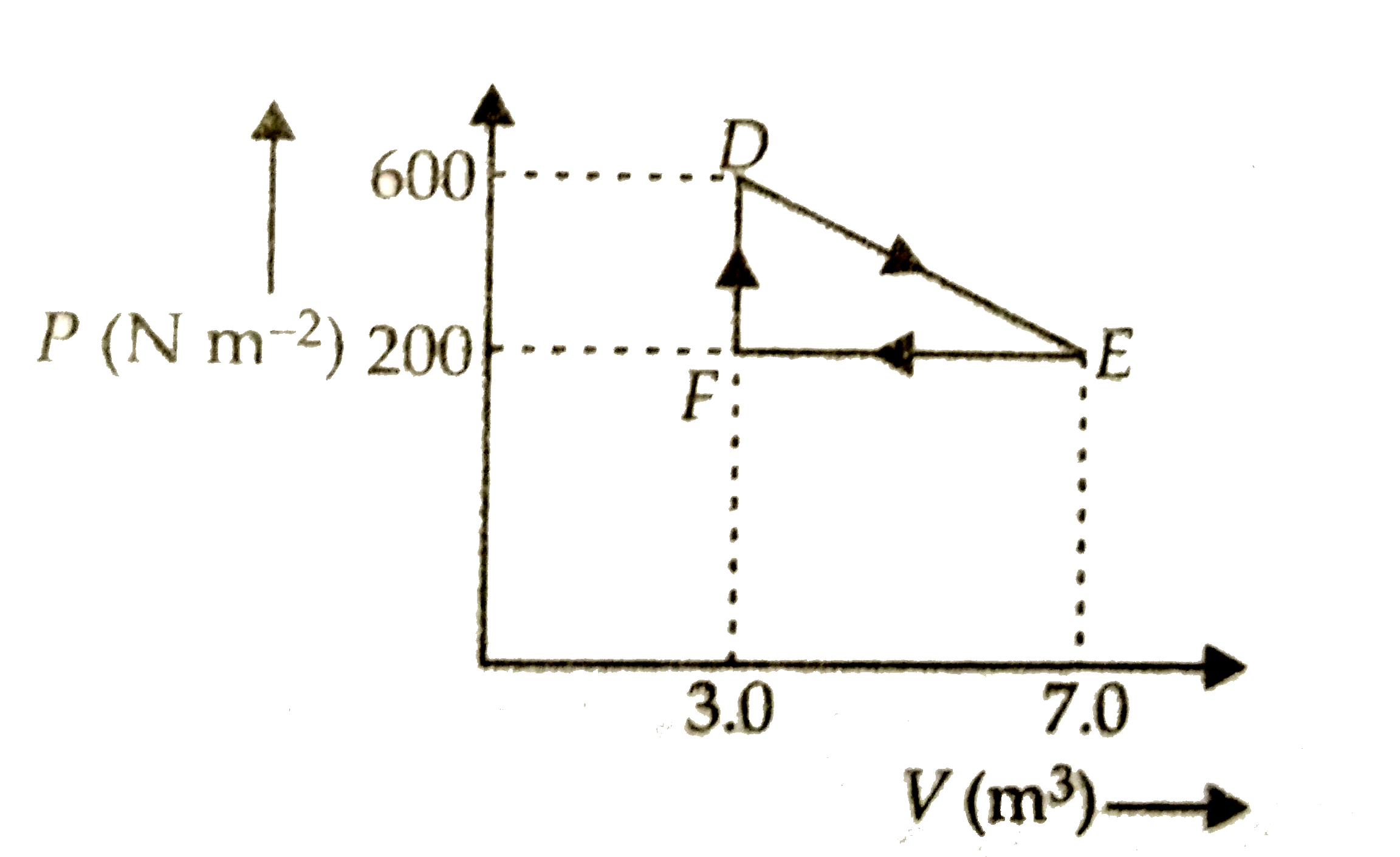
- a thermodynamic process is carried out from an original state D to an ...
Text Solution
|
- A thermodynamic system is taken from an original state D to an interme...
Text Solution
|
- a thermodynamic process is carried out from an original state D to an ...
Text Solution
|
- किसी उष्मागतिकीय निकाय को मूल अवस्था से मध्यवर्ती अवस्था तक चित्र में ...
Text Solution
|
- किसी ऊष्मागतिकीय निकाय को मूल अवस्था से मध्यवर्ती अवस्था तक चित्र में ...
Text Solution
|
- किसी ऊष्मागीतकीय निकाय को मूल अवस्था से मध्यवर्ती अवस्था तक चित...
Text Solution
|
- A thermodynamic system is taken from original state D to an intermedia...
Text Solution
|
- A thermodynamic system is taken from an original state to an intermedi...
Text Solution
|
- একটি তাপগতীয় সংস্থাকে প্রাথমিক অবস্থা D থেকে অন্তর্বর্তী অবস্থা E-তে ...
Text Solution
|