Similar Questions
Explore conceptually related problems
Recommended Questions
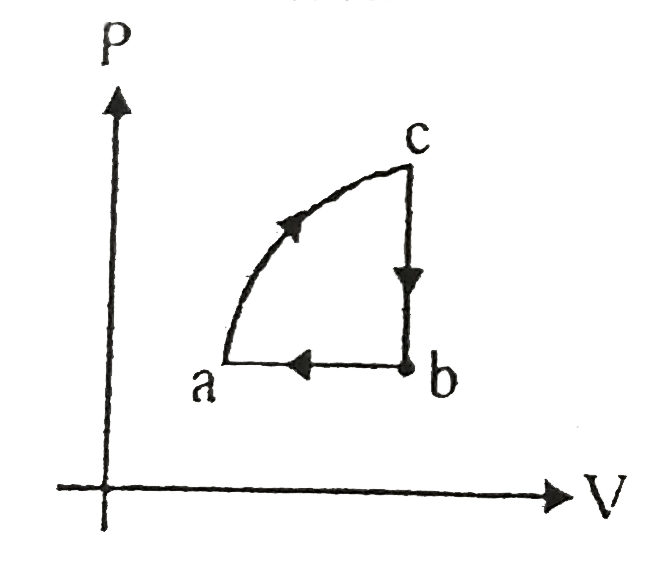
- In the given cyclic process from c to b 40 J heat is exchanged from b ...
Text Solution
|
- In the given cyclic process from c to b 40 J heat is exchanged from b ...
Text Solution
|
- A system works in a cyclic process. It absorbs 20 calories of heat and...
Text Solution
|
- If change in internal energy -80 J. The work done by system is +40 J. ...
Text Solution
|
- An ideal gas undergoes a cyclic process as shown. Part of the process ...
Text Solution
|
- IF 900 J/g of heat is exchanged at boiling point of water then increas...
Text Solution
|
- The work done by a system is 8 J, when 40 J heat is supplied to it. Th...
Text Solution
|
- A heat engine is involved with exchange of heat of 1915 J , - 40 J , +...
Text Solution
|
- In a cyclic process shown in the figure an ideal gas is adiabatically ...
Text Solution
|