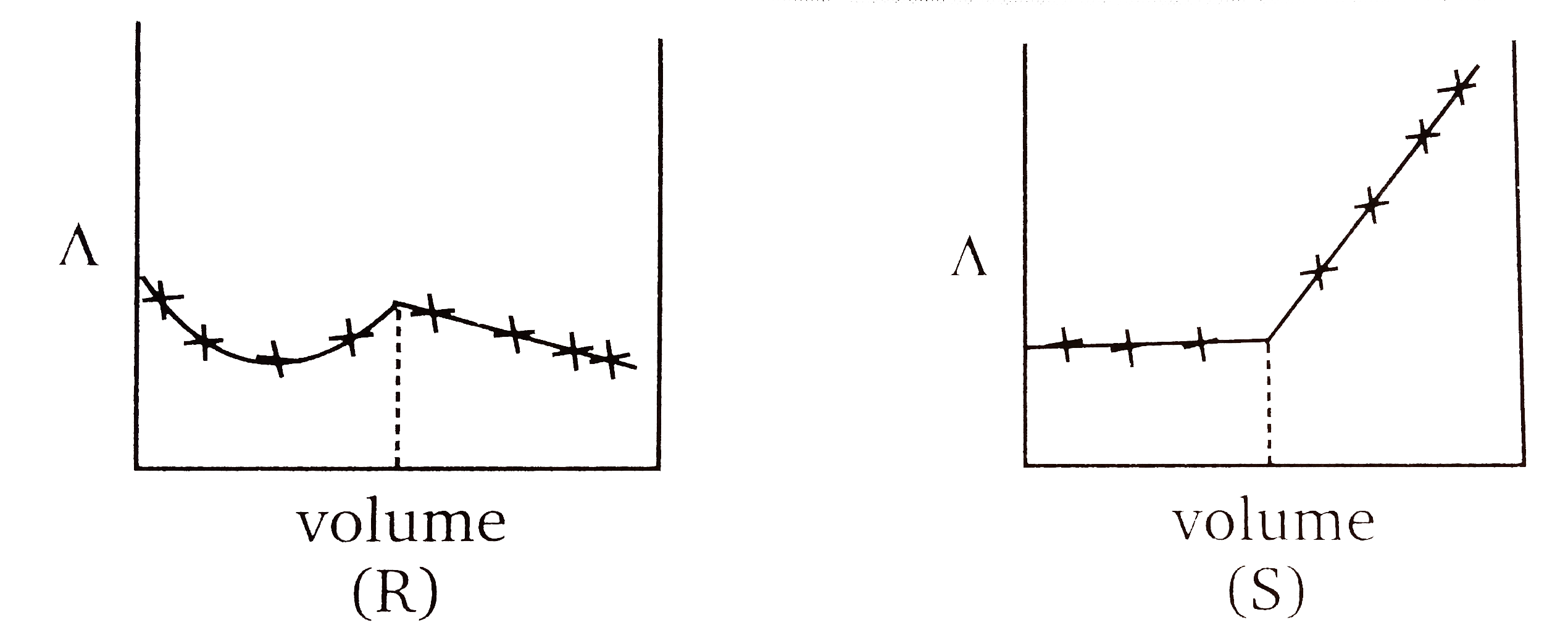
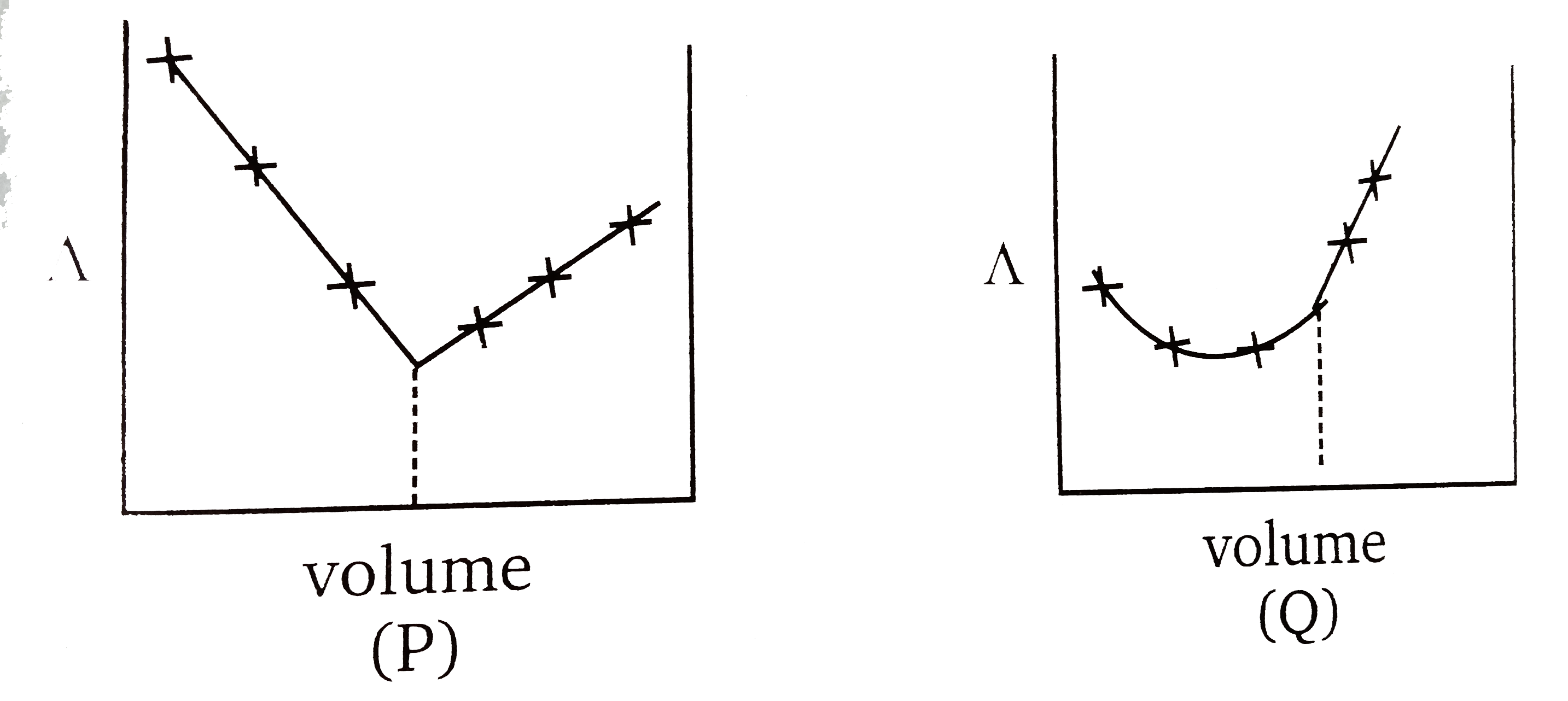A
B
C
D
Text Solution
Verified by Experts
The correct Answer is:
Topper's Solved these Questions
Similar Questions
Explore conceptually related problems
ALLEN-ELECTROCHEMISTRY-EXERCISE -05 [B]
- Tollen reagent is used for the detection of aldehydes. When a solution...
Text Solution
|
- Tollen reagent is used for the detection of aldehydes. When a solution...
Text Solution
|
- Tollen reagent is used for the detection of aldehydes. When a solution...
Text Solution
|
- Chemical reactions involve interation of atoms and molecules. A large ...
Text Solution
|
- Chemical reactions involve interation of atoms and molecules. A large ...
Text Solution
|
- Chemical reactions involve interation of atoms and molecules. A large ...
Text Solution
|
- Redox reactions play a pivotal role in chemistry and biology. The valu...
Text Solution
|
- Redox reactions play a pivotal role in chemistry and biology. The valu...
Text Solution
|
- Electrolysis of dilute aqueous NaCl solution was carried out by passin...
Text Solution
|
- For the reduction of NO(3)^(c-) ion in an aqueous solution, E^(c-) is ...
Text Solution
|
- The concentration of potassium ions inside a biological cell is at lea...
Text Solution
|
- The concentration of potassium ions inside a biological cell is at lea...
Text Solution
|
- Consider the following cell reaction : 2Fe(s)+O(2)(g)+4H^(o+)(aq) ra...
Text Solution
|
- AgNO(3)(aq.) was added to an aqeous KCl solution gradually and the con...
Text Solution
|
- The electrochemical cell shown below is a concentration cell. M|M^(2+)...
Text Solution
|
- The electrochemical cell shown below is a concentration cell. M|M^(2+)...
Text Solution
|
- The standard reduction potential data at 25^(@)C is given below E^(@...
Text Solution
|
- An aqueous solution of X is added slowly to an aqueous solution of Y a...
Text Solution
|
- In a galvanic cell, the salt bridge.
Text Solution
|
- For the following electrochemical cell at 298K Pt(s)+H(2)(g,1"bar") ...
Text Solution
|