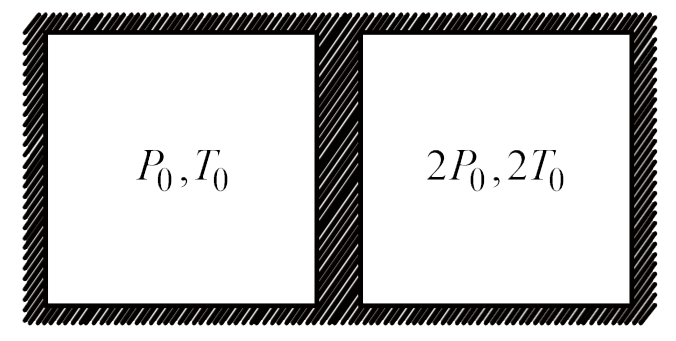A
B
C
D
Text Solution
Verified by Experts
Topper's Solved these Questions
Similar Questions
Explore conceptually related problems
DISHA-KINETIC THEORY OF GASES-physics
- In the two vessels of same volume, atomic hydrogen and helium at press...
Text Solution
|
- For gas at a temperature T the root-mean-square speed v(rms), the most...
Text Solution
|
- One mole of gas having gamma = 7//5 is mixed with 1 mole of a gas havi...
Text Solution
|
- The value of the gas constant (R) calculated from the perfect gas equa...
Text Solution
|
- Gas at a pressure P(0) in contained as a vessel. If the masses of all ...
Text Solution
|
- The mean kinetic energy per unit volume of gas (E) is related to avera...
Text Solution
|
- Mean kinetic energy (or average energy) per gm molecule of a monoatomi...
Text Solution
|
- At which of the following temperatures would the molecules of a gas ha...
Text Solution
|
- The kinetic energy of one gram molecule of a gas at normal temperature...
Text Solution
|
- 70 calories of heat required to raise the temperature of 2 moles of an...
Text Solution
|
- A vessel contains a mixture of one mole of oxygen and two moles of nit...
Text Solution
|
- From the following statements, concerning ideal gas at any given tempe...
Text Solution
|
- Let barv,v(rms) and vp respectively denote the mean speed. Root mean s...
Text Solution
|
- A gas in container A is in thermal equilibrium with another gas in con...
Text Solution
|
- A diathermic piston divides adiabatic cylinder of volume V0 into two e...
Text Solution
|
- A diathermic piston divides adiabatic cylinder of volume V0 into two e...
Text Solution
|
- A diathermic piston divides adiabatic cylinder of volume V0 into two e...
Text Solution
|
- Assertion : Internal energy of an ideal gas does not depend upon volum...
Text Solution
|
- Assetion : Equal masses of helium and oxygen gases are given equal qua...
Text Solution
|
- Statement-1 : Maxwell speed distribution graph is asymmetric about ...
Text Solution
|