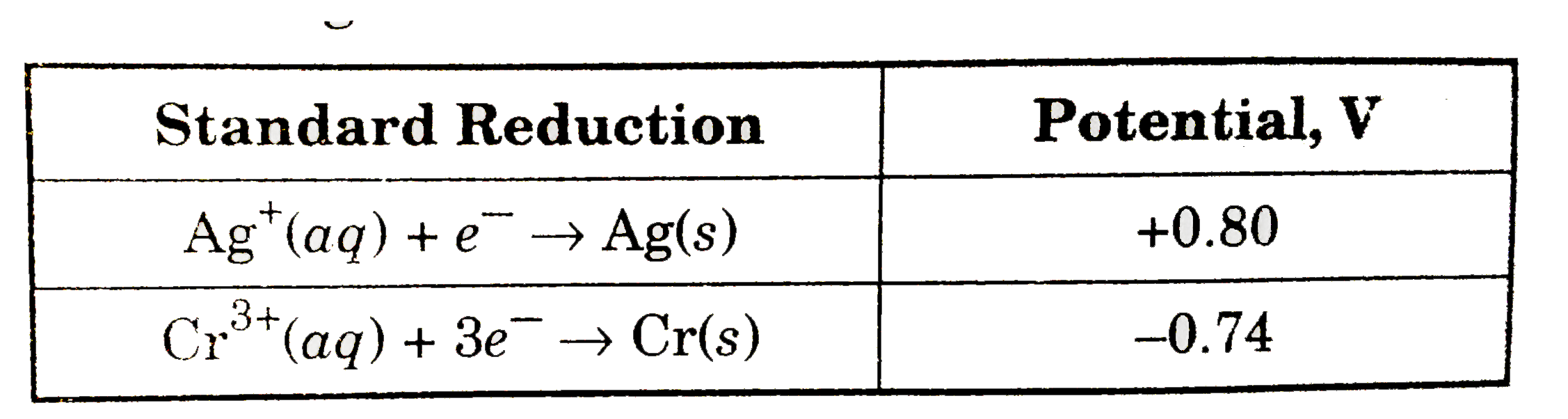A
B
C
D
Text Solution
Verified by Experts
The correct Answer is:
Topper's Solved these Questions
ELECTROCHEMISTY
GRB PUBLICATION|Exercise Faraday s Laws and Electrolysis|87 VideosELECTROCHEMISTY
GRB PUBLICATION|Exercise Conductance and Kohlrausch s law|40 VideosELECTROCHEMISTY
GRB PUBLICATION|Exercise Batteries|10 VideosD-BLOCK ELEMENTS
GRB PUBLICATION|Exercise Subjective Type|18 VideosENVIRONMENTAL CHEMISTRY
GRB PUBLICATION|Exercise Straight objective type|40 Videos
Similar Questions
Explore conceptually related problems
GRB PUBLICATION-ELECTROCHEMISTY-Thermodynamics in Electrochemistry
- DeltaG^(@) of the cell reaction AgCl(s) + 1/2H(2) rightarrow Ag(s) +...
Text Solution
|
- The emf of the cell Zn|ZnCl(2) (0.05M) | AgCl(s),Ag is 1.015 V at 298 ...
Text Solution
|
- The Gibbs energy for the decomposition of Al(2)O(3) at 500^(@)C is as...
Text Solution
|
- Calculate the cell EMF in mV for Pt|H(2)(1atm)|HCl(0.01M)||AgCl(s)|A...
Text Solution
|
- What is cell entropy change of the following cell? Pt(s)|H(2)(g)|CH(3)...
Text Solution
|
- At 298 K the standard free energy of formation of H(2)O(l) is -237.20 ...
Text Solution
|
- The potential of the Daniel cell, Zn |(ZnSO(4))||(CuSO(4)|Cu was repor...
Text Solution
|
- Using the data in the preceding problem, calcualte the equilibrium con...
Text Solution
|
- DeltaG = DeltaH - TDeltaS and DeltaG + T[(d(DeltaG))/(dT)](p) then ((d...
Text Solution
|
- DeltaG is the available energy (energy produced) during the electochem...
Text Solution
|
- At 298 K the standard free energy of formation of H(2)O (l) is -237.20...
Text Solution
|
- DeltaG^(@) of the cell reaction AgCl(s) + 1/2H(2) rightarrow Ag(s) +...
Text Solution
|
- The EMF of an electrolytic cell does not depend upon:
Text Solution
|
- For the reaction: C(3)H(8)(g) + 5O(2)(g) rightarrow 3CO(2)(g) + 4H(2...
Text Solution
|
- Ag^(aq) + e^(-) rightarrow Ag(s) E69@) = 0.80 V Mg^(2+)(aq) + 2e^(-)...
Text Solution
|
- What is the DeltaG^(@) value for the electrochemical cell below? Cu(...
Text Solution
|
- 10Cl^(-)(aq) + 2MnO(4)^(-)(aq) + 16H^(+)(aq) rightarrow 5Cl(2)(g) + 2M...
Text Solution
|
- The standard potential for the reaction, Cl(2)(g) + 2Br^(-)(aq) righ...
Text Solution
|
- What is the approximate value of the equilibrium constant, K(eq), at 2...
Text Solution
|
- 3Ni^(2+) + 2Al rightarrow 2Al^(3+) + 3Ni, E^(@0 = 1.41 V For the rea...
Text Solution
|