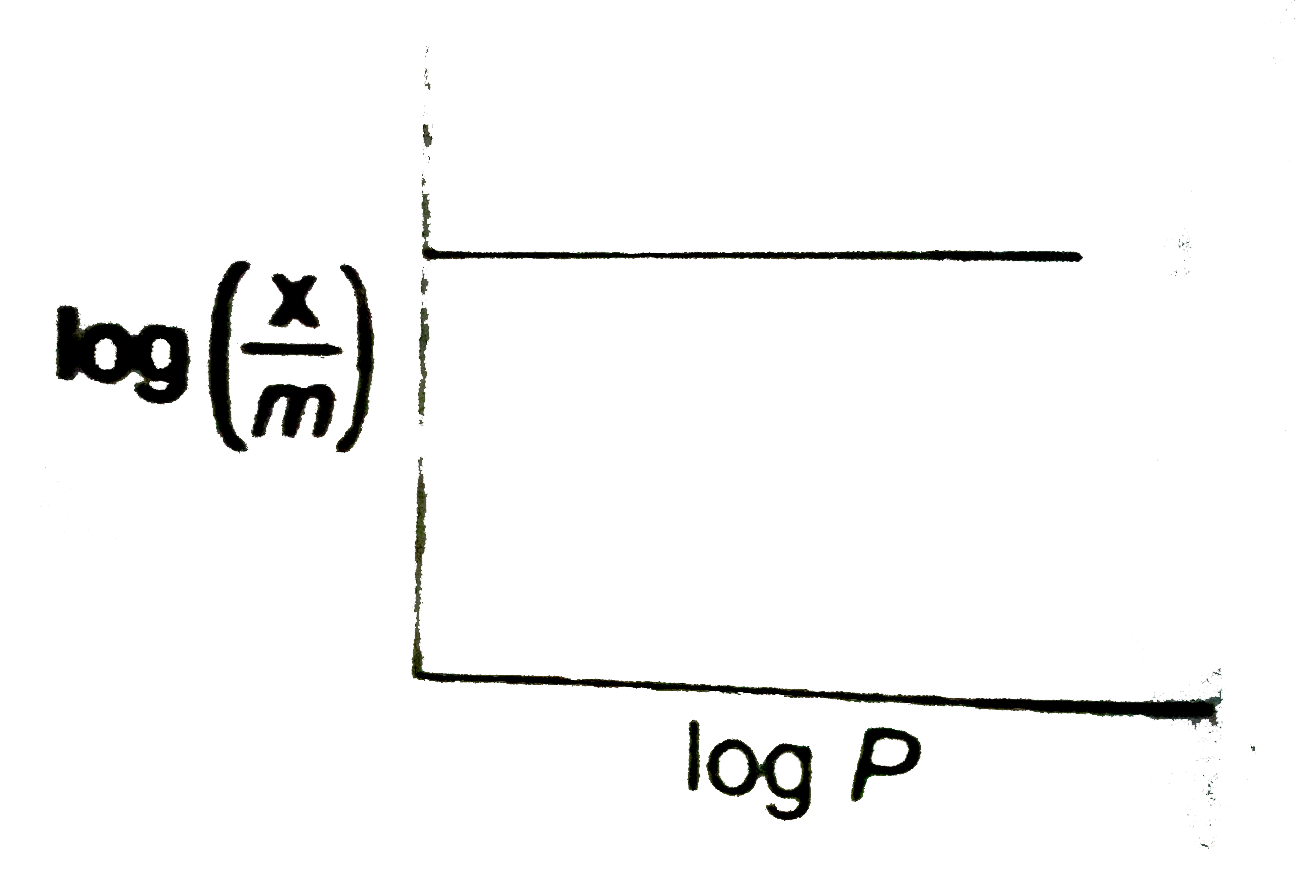A group between x/m and the presure P of the gas at a constant temperature is called adsorption siotherm. Where x is the no. of moles of the adsorbate and m is the mass of the adsorbent. adsoption isotherms of different shaopes have been experimentally observed .. According to frundlich adosroption isthem,
`x//m = KP^(1//n)`
where K and N are constant paraments depending upon the nature of the solid and gas
Adsoption isothem of log `(x/m)` and log P was found of the type .