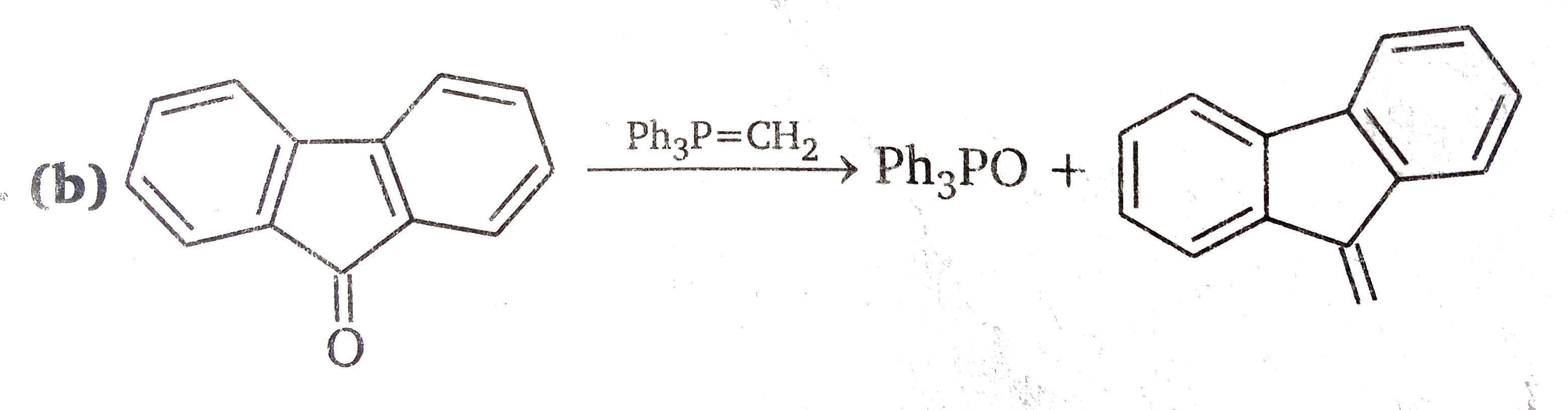
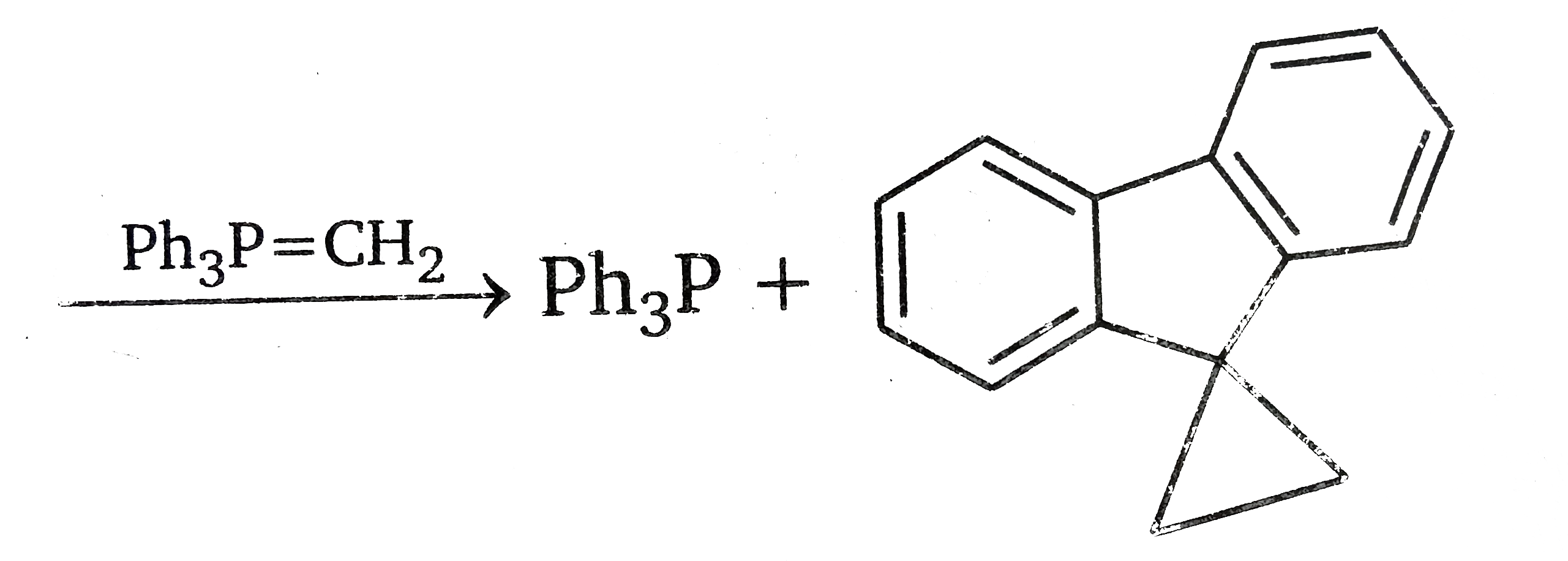
Since there is no carbonyl group in the first product, the double bond of the exocyclic methaylene gorup has reacted with methylenephosphorane. We may therefore propose a mechanism based on the assumption that the exocyclic double bond behaves like the double bond in a carbonyl group.The latter reaction is.
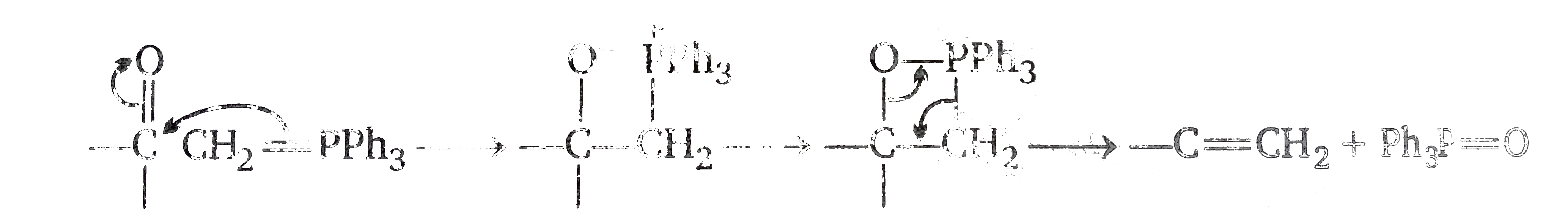
However in the case under consideration, there is no oxygen atom, phosphine and not phosphine oxide is eliminated, and a singlr and not a double bond is formed with the methtylene group. This can be accomodatd by replacing `O^(-)` by `gt C^(-)` (of the five- membered ring). and then as follows.
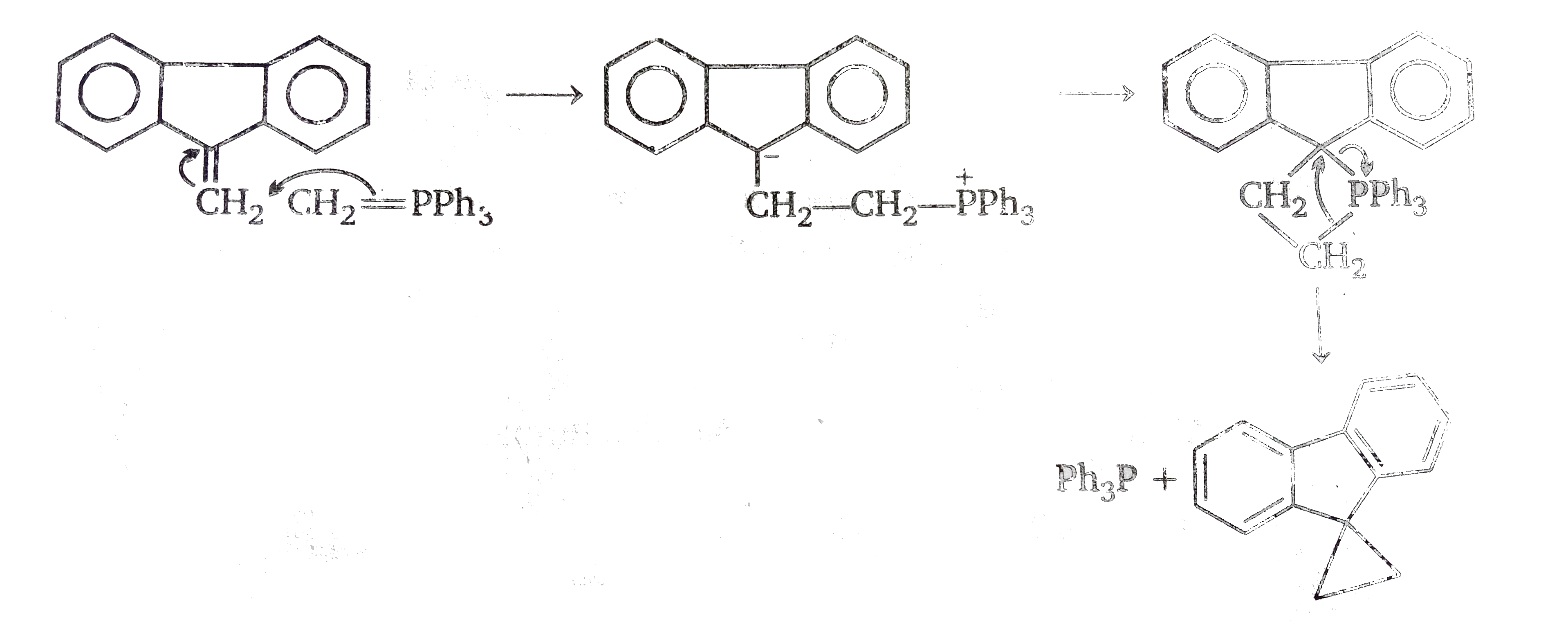
In this sequence,we have propopsed a complete parallel with the carbonyl group and this results in the formation of a spir-compound conataing phosphours in one ring. The final prodced is also a spiro-compound, but since this conatins a three memerbed ring. it could be argued that this is less stable than is precuros - which contains a four -membered the mechanism involving neighbourng group participation i.e.
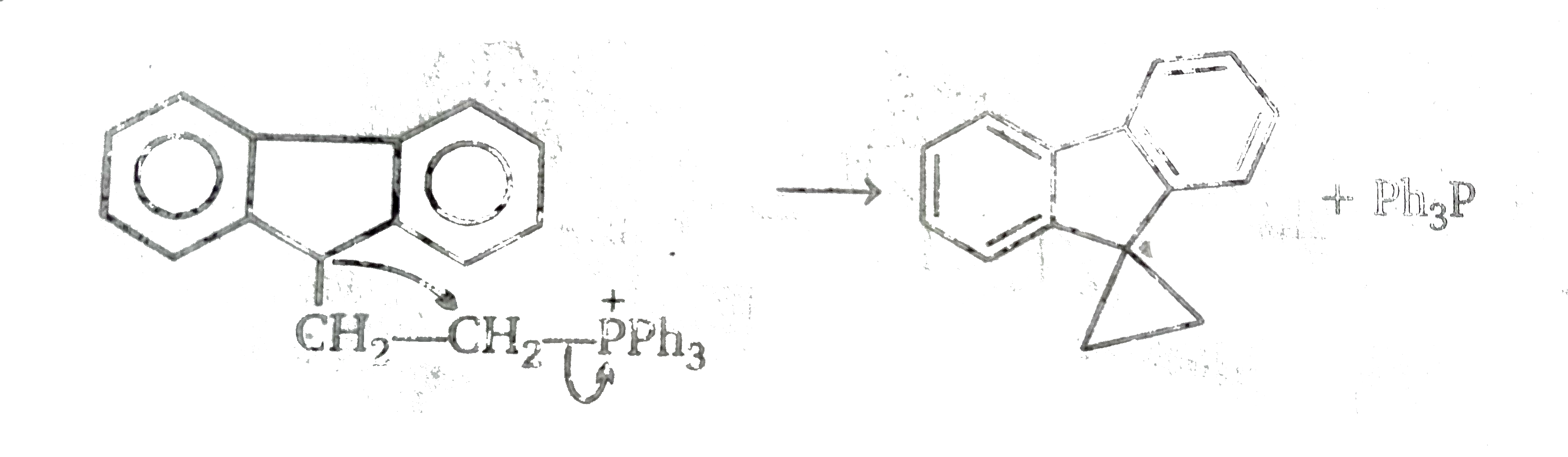
This route would be energetically more favourable than the other one and so is more likely one.