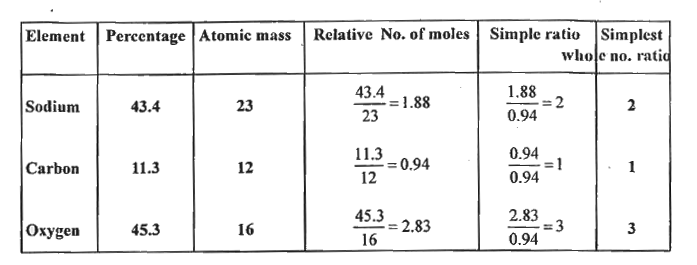
Text Solution
Verified by Experts
Topper's Solved these Questions
Similar Questions
Explore conceptually related problems
SUBHASH PUBLICATION-SOME BASIC CONCEPTS OF CHEMISTRY -NUMERICAL PROBLEMS AND ANSWERS:
- Find the Molarity of Hydrochloric acid containing 31.5% of hydrochlori...
Text Solution
|
- Calculate the molecular mass of (i) oxalic and (ii) Formic acid (iii) ...
Text Solution
|
- 25.0 cm^(3) of an acid required exactly 20.5 cm^(3) of deci molar base...
Text Solution
|
- Exactly 20.0 cm^(3) of nitric acid neutralized 28.4 cm^(3) of 0.25 M N...
Text Solution
|
- 18.5 cm^(3) of oxalic acid was completely neutralised by 20.0 cm^(3), ...
Text Solution
|
- Calculate the number of moles of atoms in 10.2 g of sodium.
Text Solution
|
- Calculate the number of moles in (a) 10 g of Hydrogen molecules (b) 30...
Text Solution
|
- Calculate the mass of the following is grams (a) 5.4 moles of O(2) (b)...
Text Solution
|
- How many atoms of oxygen are present in 300 g of CaCO(3)?
Text Solution
|
- Calculate number of atoms in (a) 2.5 mole atoms of nitrogen (b) 8.4 of...
Text Solution
|
- Calculate the number of molecules in each of the following (a) 24 g of...
Text Solution
|
- A substances on analysis for the following percentage composition the ...
Text Solution
|
- A compound has the following composition Mg = 9.76%, S = 13.01 %, O=26...
Text Solution
|
- A compound has the following percentage composition. Carbon 80%, Hydro...
Text Solution
|
- An organic compound on analyis found to contain 92.3 % carbon and 7.7%...
Text Solution
|
- 4.2 g of Mg is burnt in 4.8 g of slushy to form magnesium sulphide.Wha...
Text Solution
|
- 50.0 kg of N(2) (g) and 10.0 kg of H(2) (g) are mixed, to produce NH(3...
Text Solution
|
- Commercially available conc. HCl contains 38% HCl by mass. (a) What is...
Text Solution
|
- How many grams of oxgyen (O(2)) are required to completely react with ...
Text Solution
|
- What is the volume of oxygen at STP can be produced by 8.1 g of potass...
Text Solution
|