Text Solution
Verified by Experts
Similar Questions
Explore conceptually related problems
Recommended Questions
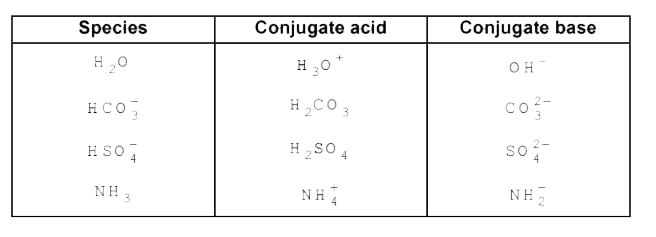
- The species H(2)O , HCO(3)^(-), HSO(4)^(-) and NH(3) and act both as B...
Text Solution
|
- The species H(2)O, HCO(3)^(Θ), HSO(4)^(Θ), NH(3) can act both as Brddo...
Text Solution
|
- The species: H(2)O, HCO(3)^(Θ), HSO(4)^(Θ) and NH(3) can act both as B...
Text Solution
|
- The species H(2)O , HCO(3)^(-), HSO(4)^(-) and NH(3) and act both as B...
Text Solution
|
- The species : H(2)O,HCO(3)^(-),HSO(4)^(-) and NH(3) can act both as Br...
Text Solution
|
- स्पीशीज H(2)O, HCO(3)^(-), HSO(4)^(-) एवं NH(3) ब्रोनस्टेड अम्ल तथा क्...
Text Solution
|
- स्पीशीज H(2)O,HCO(3)^(-),HSO(4)^(-) तथा NH(3) ब्रोंसटेड अम्ल तथा क्ष...
Text Solution
|
- The species H(2)O,HCO(3)^(-),HSO(4)^(-) and NH(3) can act both as Bron...
Text Solution
|
- The species: H(2)O,HCO(3)^(-),HSO(4)^(-) and NH(3) can act both as Bro...
Text Solution
|