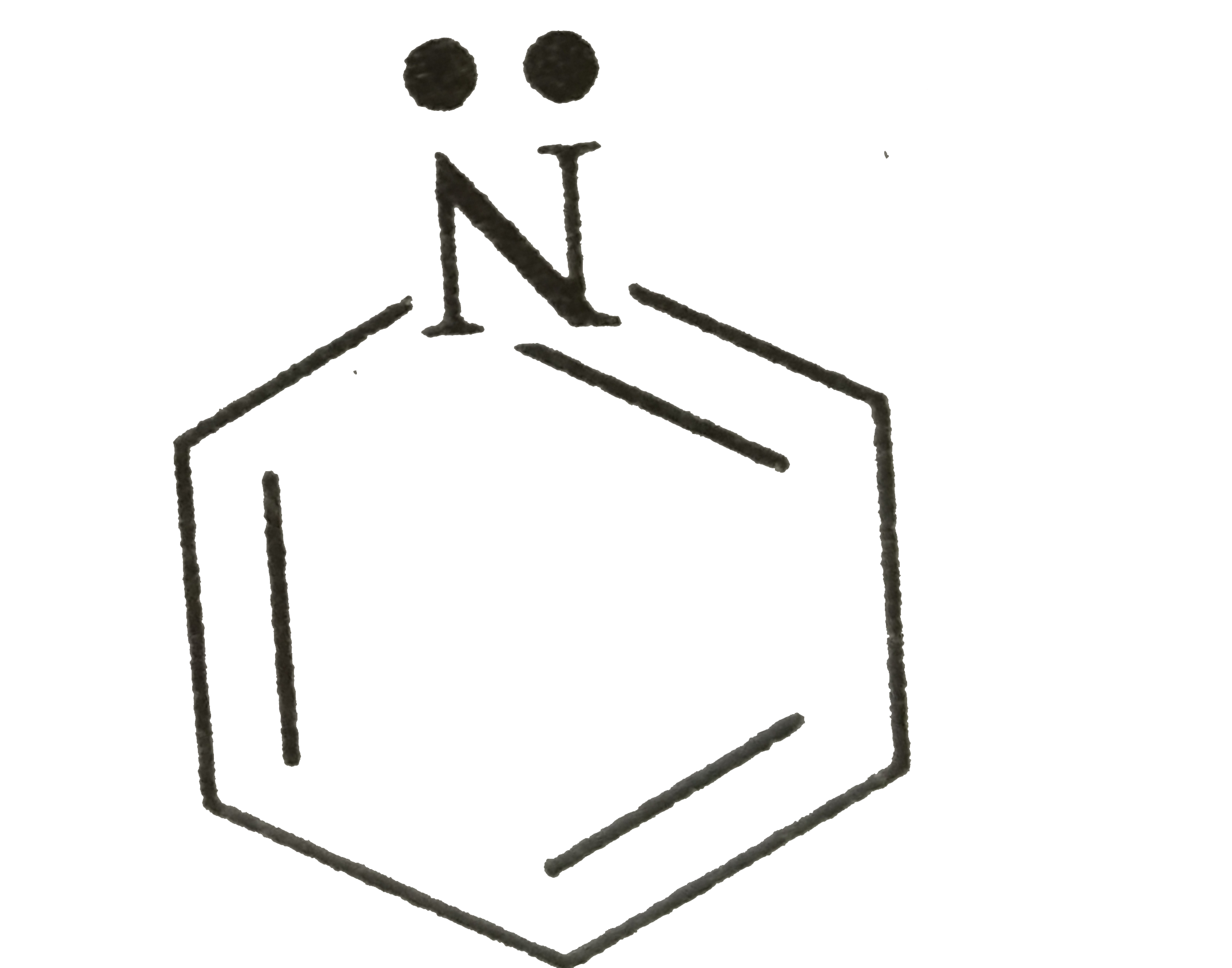
A
B
C
D
Text Solution
Verified by Experts
The correct Answer is:
Similar Questions
Explore conceptually related problems
Recommended Questions
- For any compound to be aromatic, compound should follow a given set...
Text Solution
|
- For any compound to be aromatic, compound should follow a cartain rule...
Text Solution
|
- For any compound to be aromatic, compound should follow a cartain rule...
Text Solution
|
- For any compound to be aromatic, compound should follow a given set of...
Text Solution
|
- For any compound to be aromatic, compound should follow a given set of...
Text Solution
|
- For any compound to be aromatic, compound should follow a given set...
Text Solution
|
- For any compound to be aromatic, compound should follow a given set...
Text Solution
|
- Which of the following compounds are aromatic according to Huckel's ru...
Text Solution
|
- Which of the following compounds are aromatic accroding to Huckel's ru...
Text Solution
|