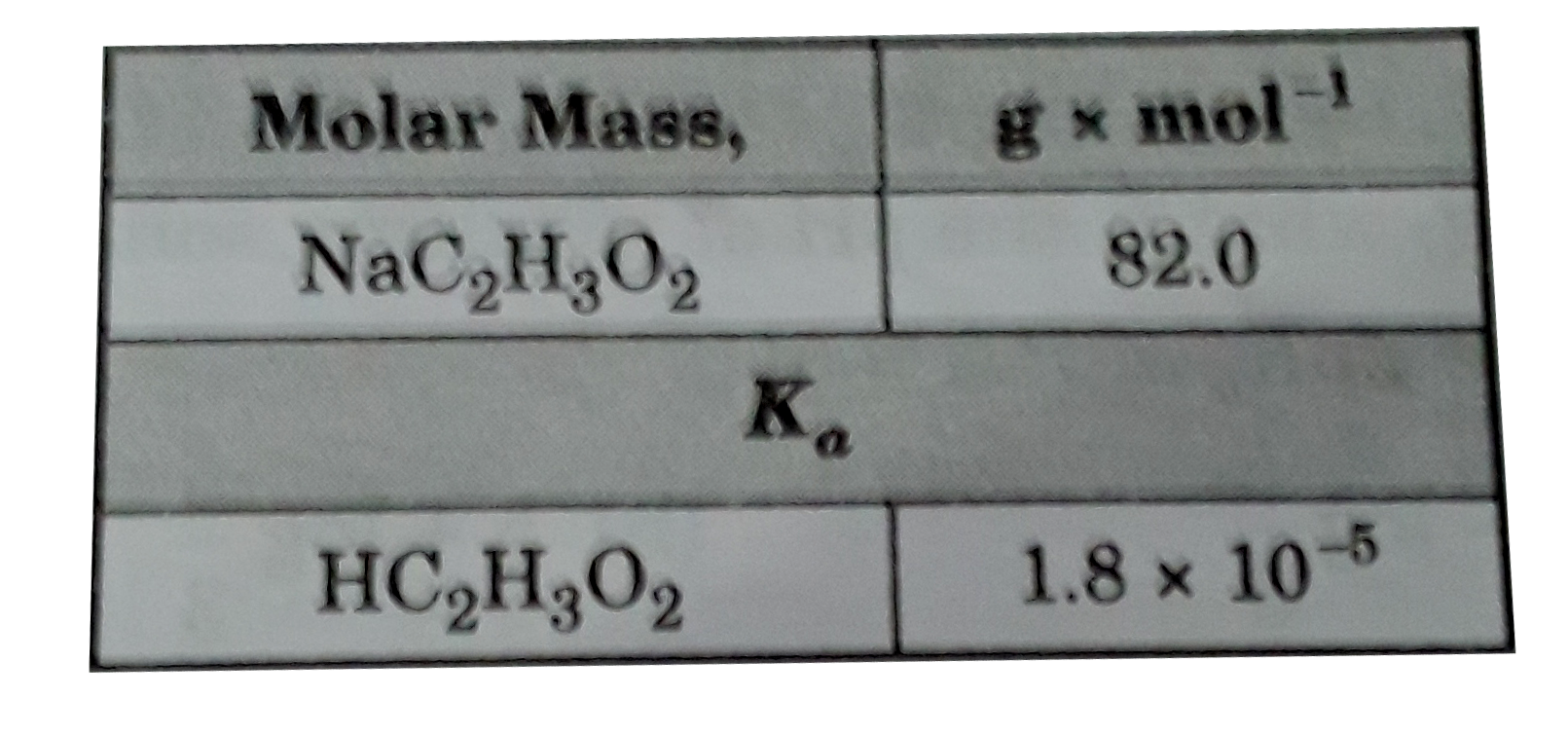Similar Questions
Explore conceptually related problems
Recommended Questions
- What is the pH of a solution made by adding 0.41 g of NaC2H3O2 to 100 ...
Text Solution
|
- What will be the pH of a solution formed by mixing 40 ml of 0.10 M HCl...
Text Solution
|
- A 75 mL solution that is 0.10 M in HC2H3O2 and 0.10 M in NaC2H3O2 has ...
Text Solution
|
- What is the pH of a solution made by adding 0.41 g of NaC2H3O2 to 100 ...
Text Solution
|
- Which of these mixtures constitute buffer solutions ? Mixture 1 : 25...
Text Solution
|
- When 0.0030 mol of HCI is added to 100 mL of a 0.10 M solution of a we...
Text Solution
|
- What will be the pH of a solution formed by mixing 40 ml of 0.10 M HCl...
Text Solution
|
- Calculate the pH of a 0.10 M ammonia solution. Calculate the pH after ...
Text Solution
|
- What will be the pH of the resulting solution if to a 100 ml of HCl so...
Text Solution
|