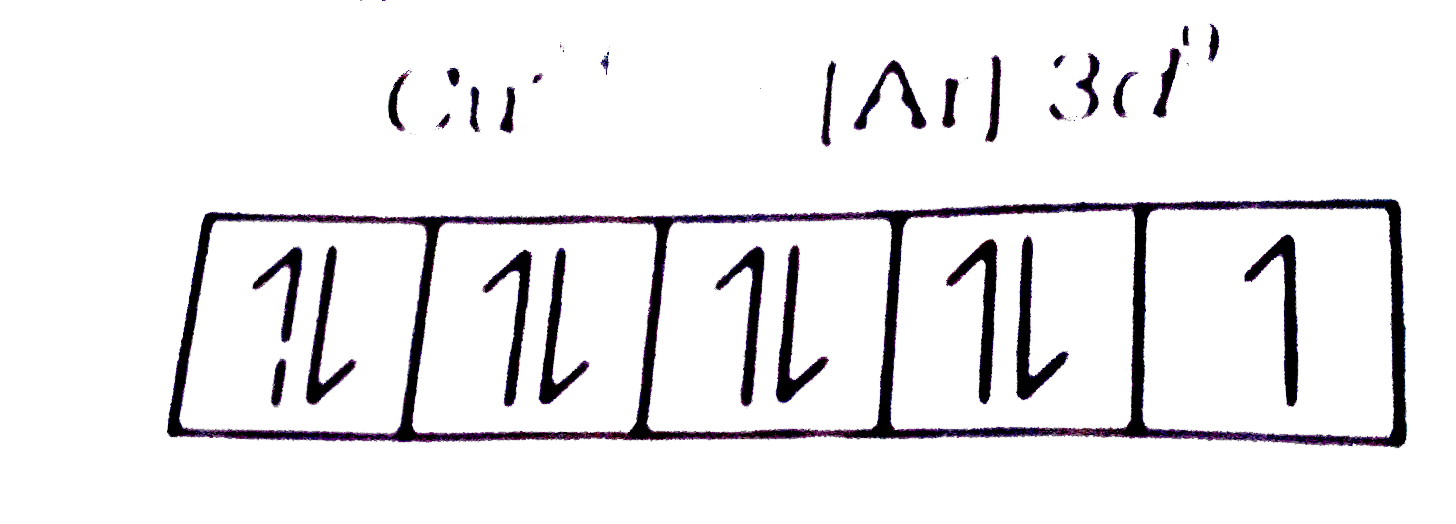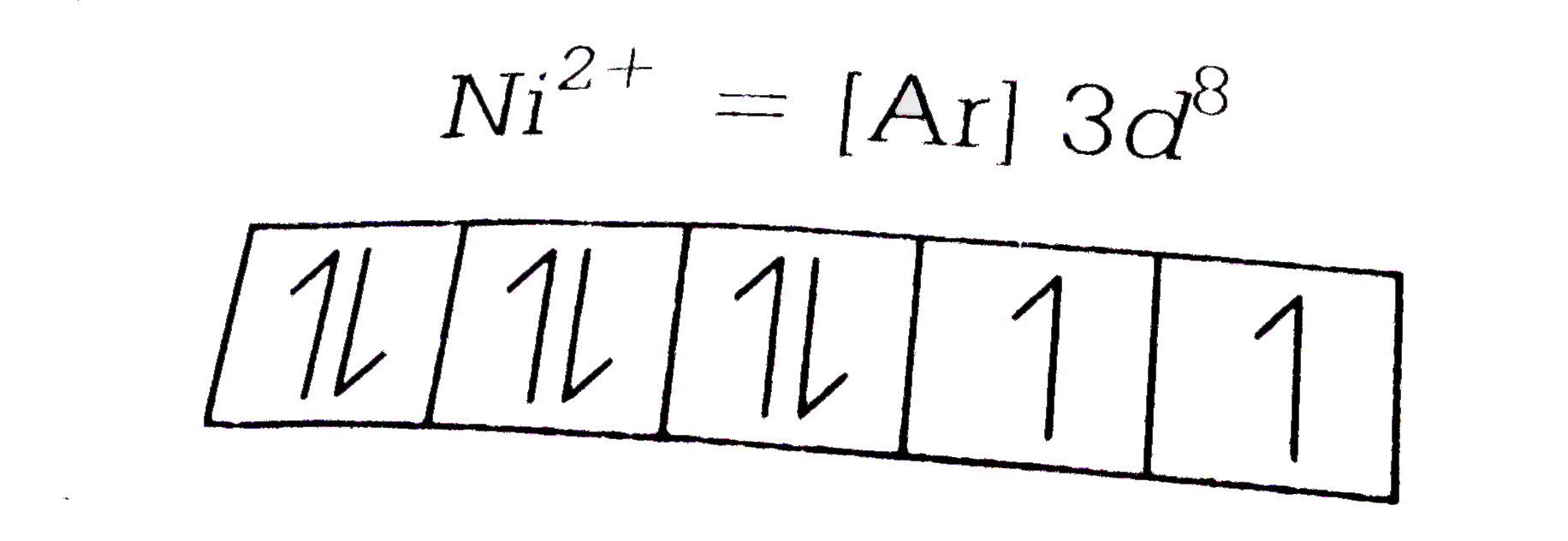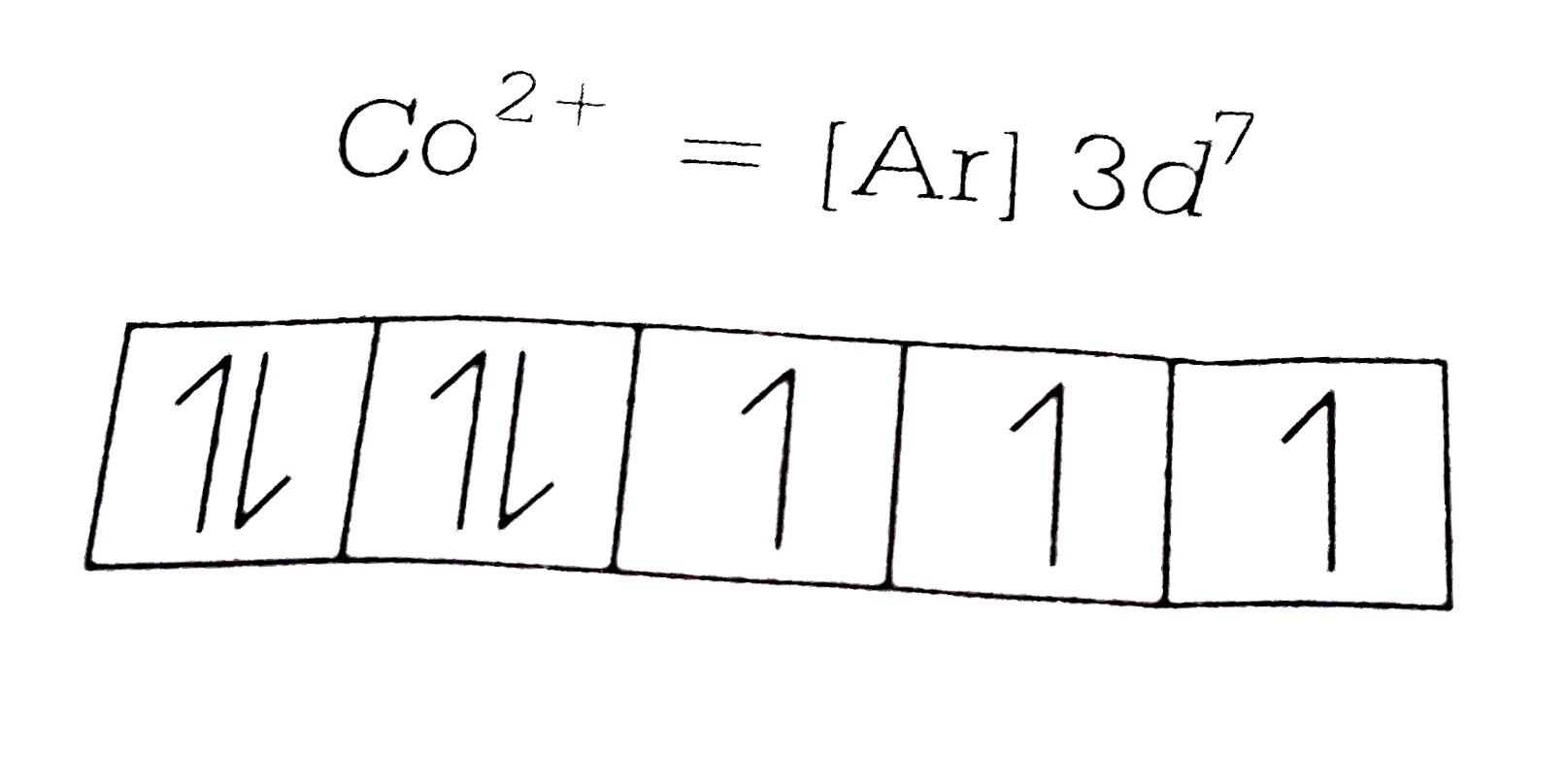A
B
C
D
Text Solution
Verified by Experts
The correct Answer is:
Topper's Solved these Questions
Similar Questions
Explore conceptually related problems
NEET PREVIOUS YEAR (YEARWISE + CHAPTERWISE)-COORDINATION COMPOUNDS-EXERCISE
- Among the following complexes, the one which shows zero crystal field ...
Text Solution
|
- The complex used as an anticancer agent is
Text Solution
|
- A magnetic moment of 1.73 B.M. will be shown by one among the followin...
Text Solution
|
- Which one of the following is an outer orbital complex and exhibits pa...
Text Solution
|
- Of the following complex ions, which is diamagnetic in natures?
Text Solution
|
- The complex [Co(NH(3))(6)][Cr(CN)(6)]" and "[Cr(NH(3))(6)][Co(CN)(6)] ...
Text Solution
|
- The complex, [Pt(py)(NH3)BrCl] will have how many geometrical isomers?
Text Solution
|
- Which of the following complex ion is not expected to absorb visible l...
Text Solution
|
- Crystal field stabilization energy for high spin d^4 octahedral comple...
Text Solution
|
- The existence of two different coloured comlexes with the composition ...
Text Solution
|
- Amongst [TiE(6)]^(2-), [CoF(6)]^(3-), Cu(2)Cl(2) and [NiCl(4)]^(2-) [A...
Text Solution
|
- Which of the following complex ions is expected to absorb visible ligh...
Text Solution
|
- Which of the following does not show optical isomerism ?
Text Solution
|
- In which of the following coordination entites the magnitude of Delta(...
Text Solution
|
- Which of the following complexes exhibits the highest paramagnetic beh...
Text Solution
|
- Which of the following will give a pair of enontiomorphs ? en = NH(2...
Text Solution
|
- The d electron congfiguration of Cr^(2+) , Mn^(2+) , Fe^(2+) and Ni^(2...
Text Solution
|
- [Cr(H2 O)6]Cl3 (at no. of Cr = 24) has a magnetic moment of 3.83 B.M. ...
Text Solution
|
- [Co(NH(3))(4)(NO(2))(2)]CI exhibits
Text Solution
|
- Which one of the following is expected to exhibit optical isomerism (e...
Text Solution
|