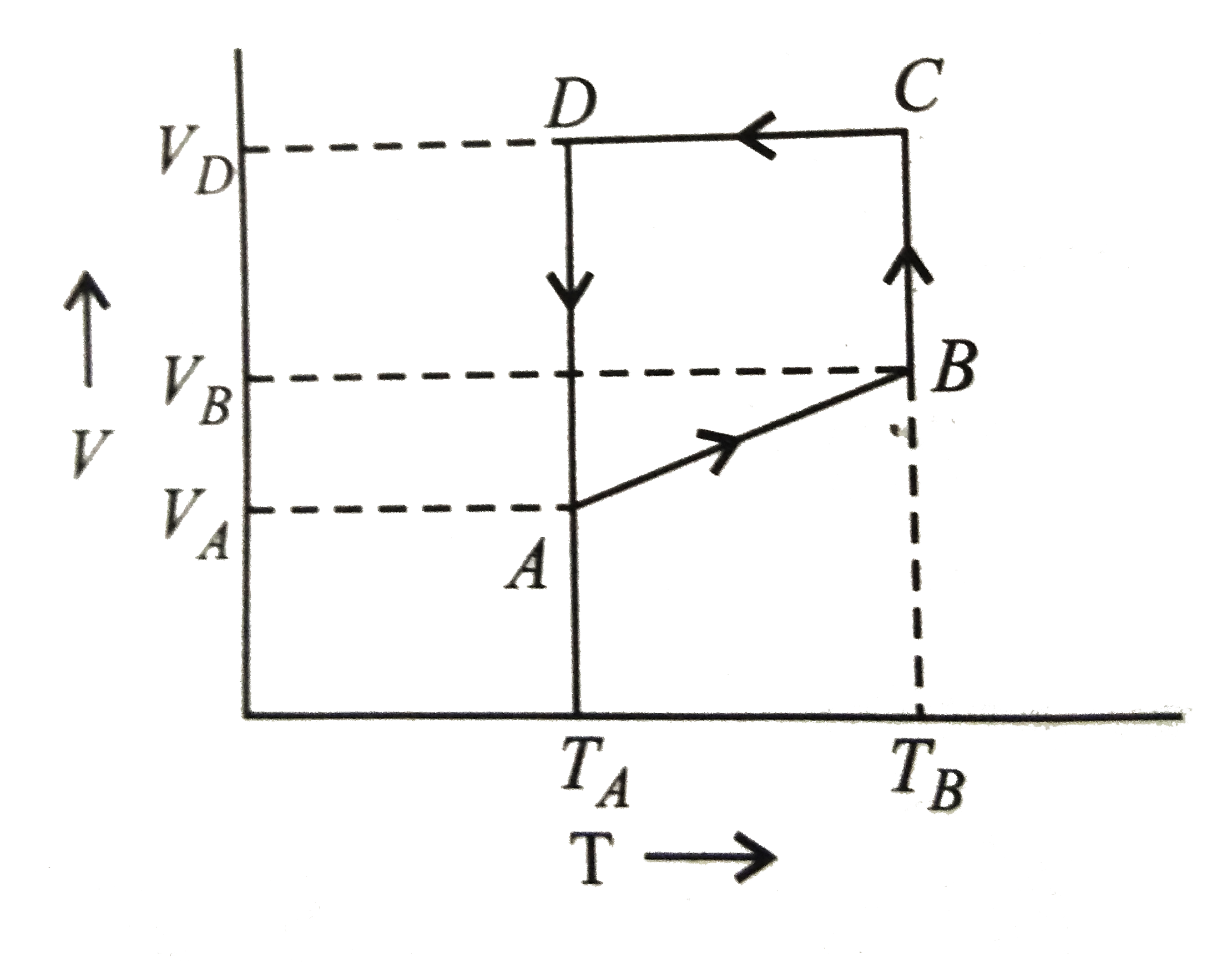
Text Solution
Verified by Experts
Topper's Solved these Questions
Chemical Thermodynamics and Thermochemistry
OP TANDON|Exercise Objective Question Level-A|177 VideosChemical Thermodynamics and Thermochemistry
OP TANDON|Exercise Set 2|18 VideosChemical Thermodynamics and Thermochemistry
OP TANDON|Exercise Miscellaneous Numerical Examples|22 VideosCHEMICAL KINETICS
OP TANDON|Exercise LINKED COMPRESHENSION TYPE QUESTIONS ( SECTION-VI)|4 VideosCLASSIFICATION AND NOMENCLATURE OF ORGANIC COMPOUNDS
OP TANDON|Exercise INTEGER|2 Videos
Similar Questions
Explore conceptually related problems
OP TANDON-Chemical Thermodynamics and Thermochemistry-Practice problems
- Calculate the entropy change for the following reaction, {:(,CaCO(3)...
Text Solution
|
- Compute the value of DeltaS at 298K for the reaction, H(2)(g)+1//2O(...
Text Solution
|
- At 300 K, the standard enthalpies of formation of C(6)H(5)COOH(s),CO(2...
Text Solution
|
- DeltaH and DeltaS for Br(2)(l) +CI(2)(g) rarr 2BrCI(g) are 29.00 k...
Text Solution
|
- For the sysnthesis of ammonia, N(2)(g)+3H(2)(g)to2NH(3)(g) Calcula...
Text Solution
|
- How much heat is required to cahgen 15.6 g of benzene from liquid into...
Text Solution
|
- Calculate the standard free energy change for the combustion of glucos...
Text Solution
|
- The specific heat at constant volume for a gas is 0.075 cal/g and at c...
Text Solution
|
- The polymerisation of ethylene to linear polyethylene is represented b...
Text Solution
|
- An athelete is given 100g of glucose (C(6)H(12)O(6)) of energy equival...
Text Solution
|
- Calculate the entropy of ideal mixing when 2 m oles of N(2), 3 moles o...
Text Solution
|
- Calculate free energy change for the reaction, H(2)(g)+Cl(2)(g)to2H-...
Text Solution
|
- For the reaction, 4C(s) +5H(2)(g) rarr nC(4)H(10)(g), DeltaH^(The...
Text Solution
|
- The temperature of a bomb calorimeter was found to rise by 1.617K when...
Text Solution
|
- 1 mol of an ideal gas is allowed to expand isothermally at 27^(@)C unt...
Text Solution
|
- One kilogram water at 0^(@)C is brought into contact with a heat reser...
Text Solution
|
- A mono-atomic ideal gas of two moles is taken through a cyclic process...
Text Solution
|
- The reaction, SOCl(2)+H(2)OtoSO(2)+2HCl, is endothermica by 49.4 kJ an...
Text Solution
|
- What amount of ice will remains when 52 g ice is added to 100 g of wat...
Text Solution
|
- Calculate the DeltaH(f)^@ of C(6)H(12)O(6)(s) from the following data:...
Text Solution
|