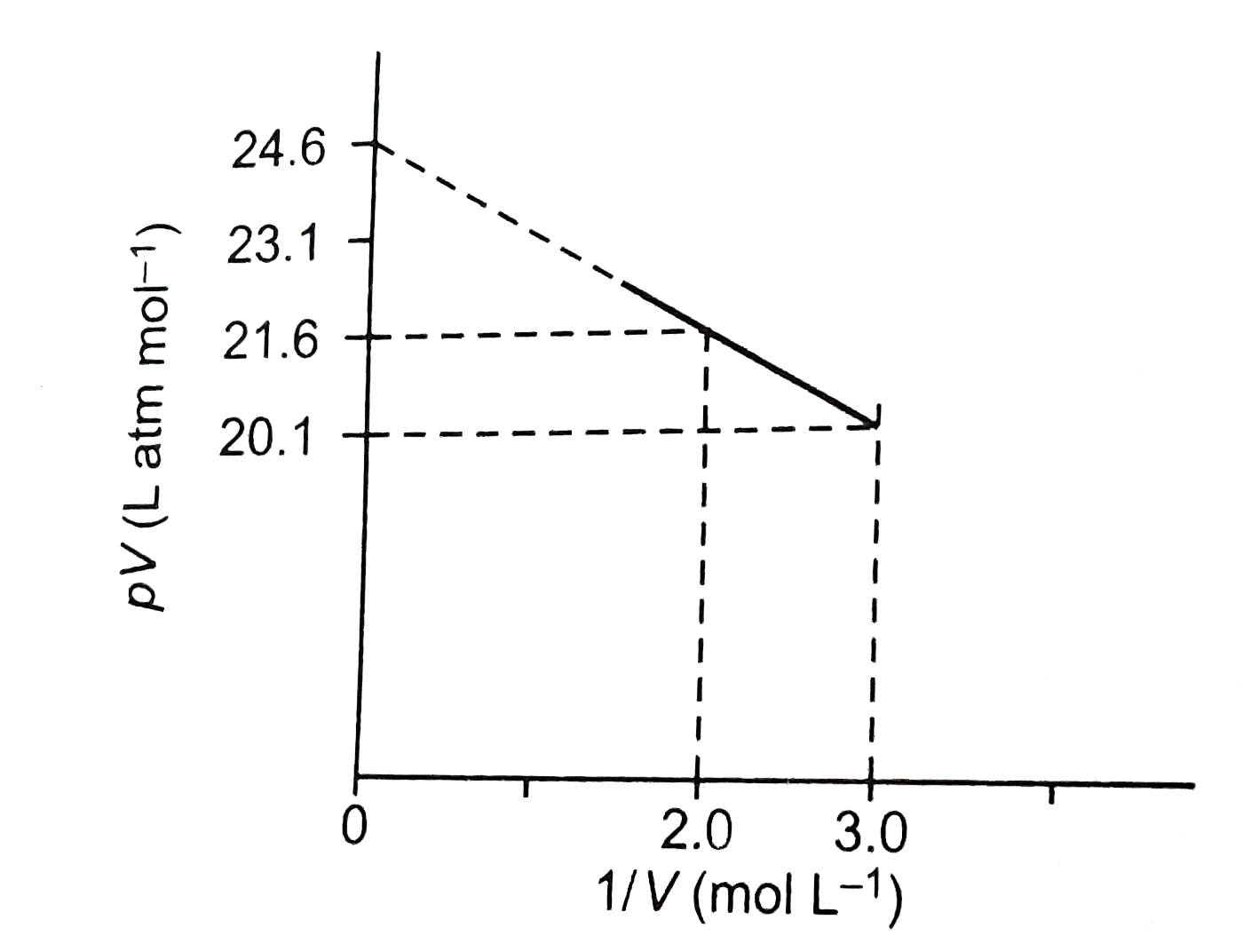
A
B
C
D
Text Solution
Verified by Experts
The correct Answer is:
Topper's Solved these Questions
GASEOUS STATE
RESONANCE|Exercise Exercise-2|1 VideosGASEOUS STATE
RESONANCE|Exercise Exercise-3|1 VideosGASEOUS STATE
RESONANCE|Exercise Exercise-1|1 VideosFUNDAMENTAL CONCEPT
RESONANCE|Exercise ORGANIC CHEMISTRY(Fundamental Concept )|40 VideosHYDROCARBON
RESONANCE|Exercise ORGANIC CHEMISTRY(Hydrocarbon)|50 Videos
Similar Questions
Explore conceptually related problems
RESONANCE-GASEOUS STATE-Exercise
- Which of the following is correct for critical temperature ?
Text Solution
|
- The vander waal gas constant 'a' is given by
Text Solution
|
- Which of the following are correct statements?
Text Solution
|
- Select the correct statements(s):
Text Solution
|
- Which is/are correct for real gases?
Text Solution
|
- Which of the following statements are incorrect?
Text Solution
|
- One of the important approach to the study of real gases involves the ...
Text Solution
|
- One of the important approach to the study of real gases involves the ...
Text Solution
|
- One of the important approach to the study of real gases involves the ...
Text Solution
|
- When pressure is increases at constant temp volume of gas decreases AB...
Text Solution
|
- When pressure is increases at constant temp volume of gas decreases AB...
Text Solution
|
- When pressure is increases at constant temp volume of gas decreases AB...
Text Solution
|
- A gas described by van der Waals equation .
Text Solution
|
- The term that corrects for the attractive forces present in a real gas...
Text Solution
|
- For one mole of a van der Waals' gas when b=0 and T=300K, the pV vs 1/...
Text Solution
|
- One mole of a monoatomic real gas satisfies the equation p(V -b) = RT ...
Text Solution
|
- In van der Waals' equation of state of the gas law the constnat 'b' is...
Text Solution
|
- a' and 'b' are van der Waals' constants for gases Chlorine is more eas...
Text Solution
|
- The compressibility factor for a real gas at high pressure is .
Text Solution
|
- If Z is a compressibility factor, van der Waals' equation at low press...
Text Solution
|