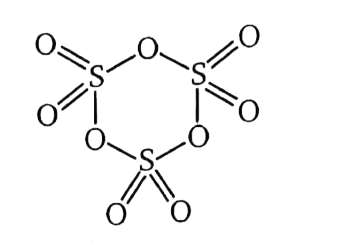
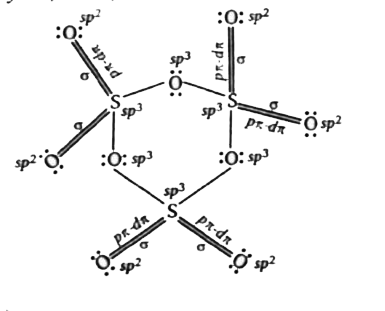
Text Solution
Verified by Experts
The correct Answer is:
Topper's Solved these Questions
Similar Questions
Explore conceptually related problems
VK JAISWAL-CHEMICAL BONDING (ADVANCED)-SUBJECTIVE PROBLEMS
- Consider the following species O(Me)(2), N(SiH(3))(3), CO, O(SiH(3))(2...
Text Solution
|
- Total number of molecules which can form H-bond among themselves. Si...
Text Solution
|
- Consider two covalent compounds AL(n(1)) and BL(n(2)), if central atom...
Text Solution
|
- Calculate the I-I distance in (Å) for given compound H(2)C(2) I(2) if ...
Text Solution
|
- There are some arrangements of atomic orbitals which are given below: ...
Text Solution
|
- Number of hybrid orbital C atoms which have 33% p-character in C(CN)(4...
Text Solution
|
- Max. no. of equal P-O bonds in P(2)O(7)^(4-) ion is :
Text Solution
|
- Consider the following species: (i) CH(3)^(+) (ii) (C(3)H(5))(3)Al...
Text Solution
|
- Calculate value of |x-y|, here x and y are the total number of bonds i...
Text Solution
|
- Consider the following compounds : (i) IF(5) " " (ii) ClI(4)^(-) "...
Text Solution
|
- Consider the following Calculate value of p+q, here p and q are tot...
Text Solution
|
- Consider the following orbitals (i)3p(x) (ii)4d(x^(2)) (iii)3d(x^(2)...
Text Solution
|
- Consider the following oxyanions : PO(4)^(3-), P(2)O(6)^(4-), SO(4)^...
Text Solution
|
- Consider the following three compounds (i)AX(2n)^(n-), (ii)AX(3n) and...
Text Solution
|
- Consider the following compounds and Calculate the value of ((P^(2)-Q^...
Text Solution
|
- Consider the following compounds (1)H(3)CF (2)H(2)CF(2) (3) CH(4) ...
Text Solution
|
- There are some species given below. (i)O(2)^(+) (ii)CO (iii)B(2) ...
Text Solution
|
- Following compounds A and B have similar structure with delocalization...
Text Solution
|
- The hybridization of central atoms of compounds A, B, C and D are sp^(...
Text Solution
|
- In compound PCl(x)F(5-x), possible values of x are 0 to 5, then calcul...
Text Solution
|