Text Solution
Verified by Experts
Topper's Solved these Questions
DUAL NATURE OF RADIATION AND MATTER
MODERN PUBLICATION|Exercise Revision Exercises (Additional Questions)|8 VideosDUAL NATURE OF RADIATION AND MATTER
MODERN PUBLICATION|Exercise Revision Exercises (Fill in the Blanks)|11 VideosDUAL NATURE OF RADIATION AND MATTER
MODERN PUBLICATION|Exercise Higher Order Thinking Skills & Advanced Level (QUESTIONS WITH ANSWERS)|8 VideosCURRENT ELECTRICITY
MODERN PUBLICATION|Exercise Chapter Practice Test|15 VideosELECTRIC CHARGES AND FIELDS
MODERN PUBLICATION|Exercise Chapter Practice Test|15 Videos
Similar Questions
Explore conceptually related problems
MODERN PUBLICATION-DUAL NATURE OF RADIATION AND MATTER -Revision Exercises (Very Short Answer Questions)
- An electron and proton are possessing the same amount of kinetic energ...
Text Solution
|
- What will be the de Broglie wavelength associated with a ball of mass ...
Text Solution
|
- What would be the de Broglie wavelength of an electron possessing 100 ...
Text Solution
|
- Plot a graph showing variation of a de Broglie wavelength (lamda) asso...
Text Solution
|
- Write the expression for the de-Broglie wavelength associated with a c...
Text Solution
|
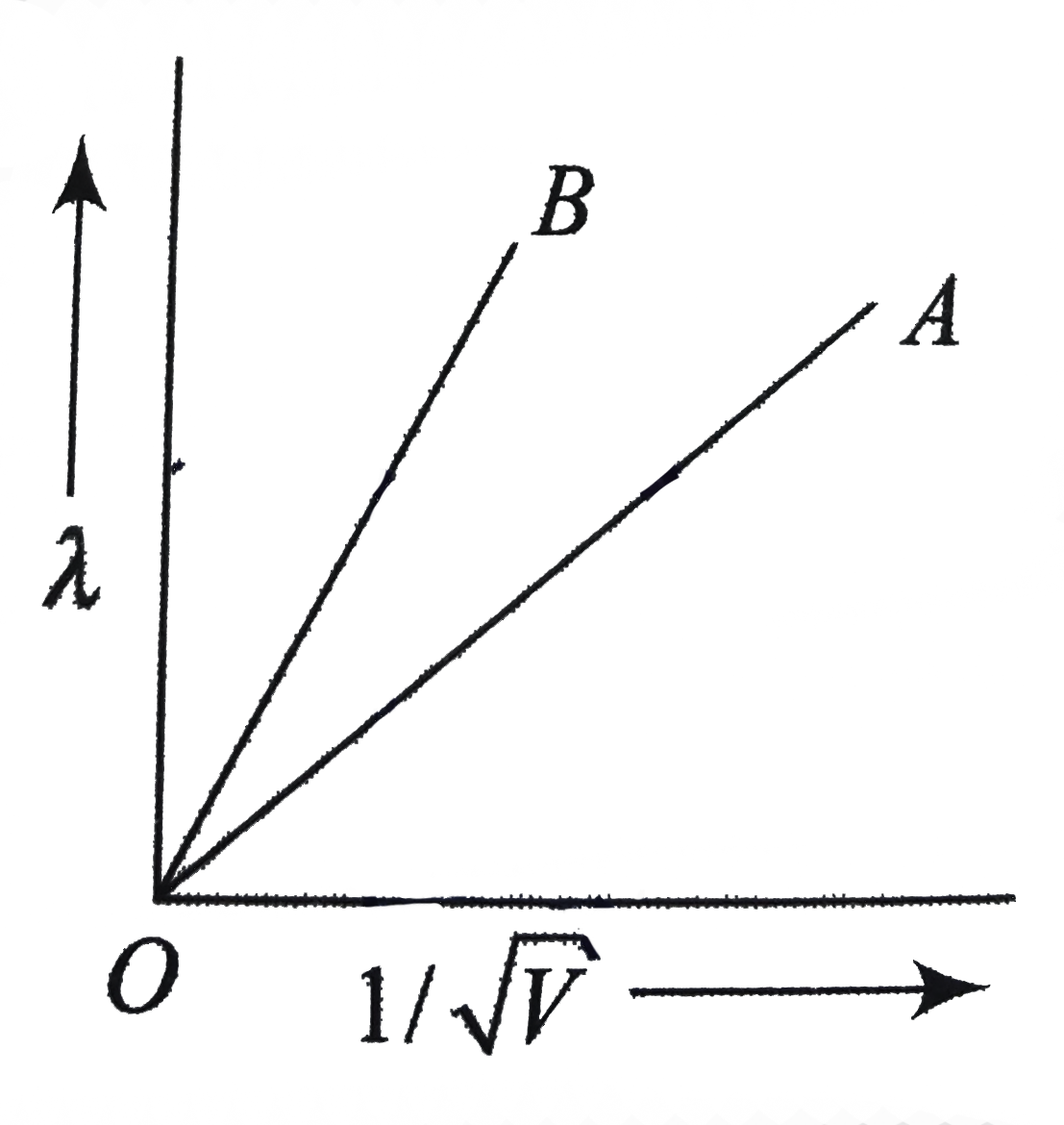
- The two lines A and B in fig. show the photo electron of de Broglie wa...
Text Solution
|
- Two metals a and b have work function 5 eV and 7 eV , respectively . W...
Text Solution
|
- Ultraviolet light is incident on two photosensitive materials having w...
Text Solution
|
- What do you mean by photoelectric efficiency ?
Text Solution
|
- Can photoelectric efficiency be greater than One (1) ?
Text Solution
|
- The de Broglie wavelength lambda of a particle
Text Solution
|
- Why are alkali metal surfaces most suited as photo-sensitive surfaces?
Text Solution
|
- Explain giving reasons for the following : (a) Photoelectric current...
Text Solution
|
- The stopping potential in an experiment on a photo electric effect is ...
Text Solution
|
- The energy that should be added to an electron, to reduce its de-Brogl...
Text Solution
|
- In Davisson - Germer experiment , find the glancing angle if the anlge...
Text Solution
|
- Plot a graph of the de - Broglie wavelength associated with electron a...
Text Solution
|
- An electron and alpha particle have the same de-Broglie wavelength as...
Text Solution
|
- Which of the following pheniomeana exhibits particle nature of light ?
Text Solution
|
- Maximum kinetic energy of photoelectron depends on
Text Solution
|