Text Solution
Verified by Experts
Similar Questions
Explore conceptually related problems
Recommended Questions
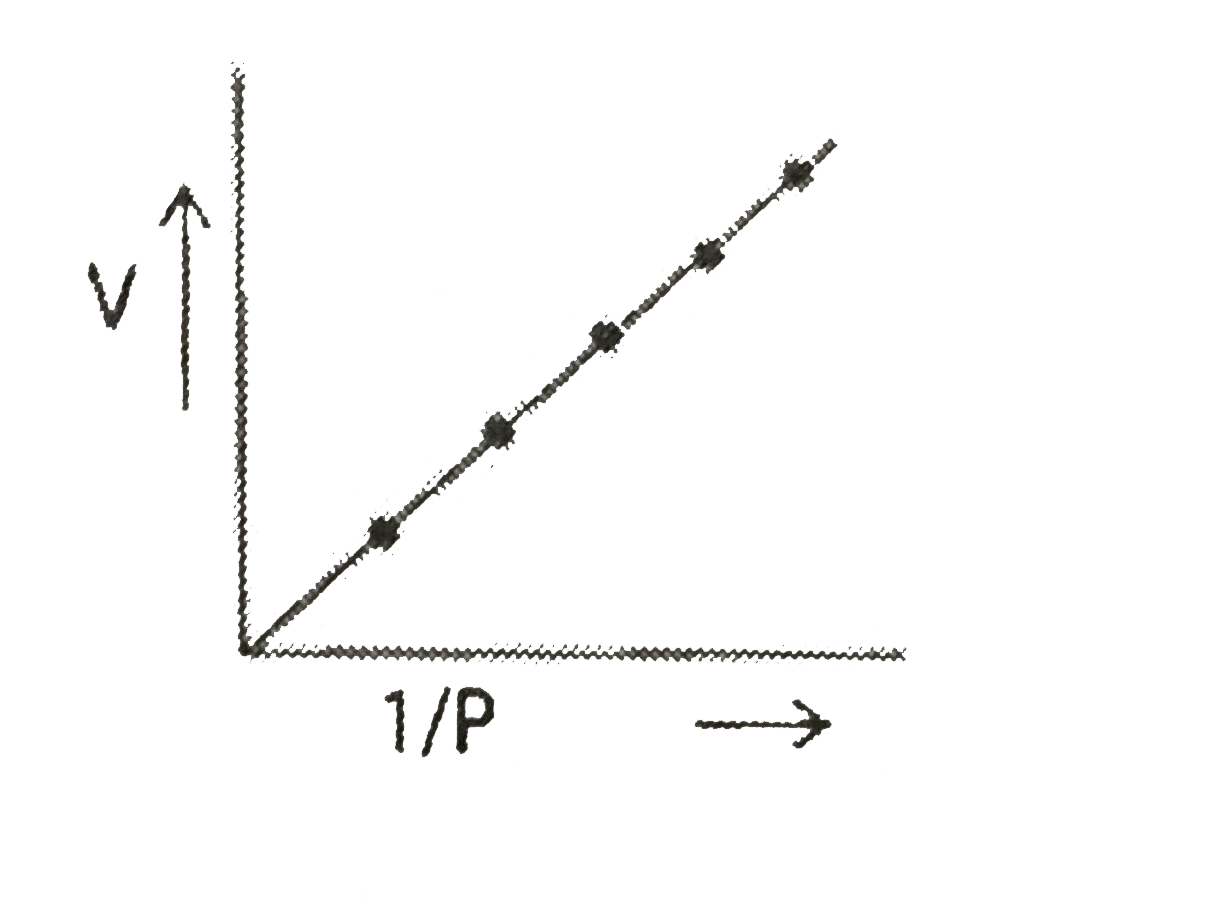
- The slope of a given straight lime graph with constant temperature is...
Text Solution
|
- Calculate the moles of an ideal gas at pressure 2 atm and volume 1 L a...
Text Solution
|
- A gas has a volume of 6.0L at a pressure of 0.80 atm. What is the volu...
Text Solution
|
- The pressure of a gas of volume 22.4 l is 3 atm at certain temperature...
Text Solution
|
- 5 L of methane gas at 2 atm pressure is compressed to 1 .6 L at contan...
Text Solution
|
- The slope of a given straight lime graph with constant temperature is ...
Text Solution
|
- The pressure of gas at 100^(@)C is 2 atm. When the gas is heated at co...
Text Solution
|
- A steel tank holds 0.2 m^3 of air at a pressure of 5 atm. The volwne o...
Text Solution
|
- Volume of a given mass of an ideal gas is VL at 27^(@)C and 1 atm pres...
Text Solution
|