Text Solution
Verified by Experts
Similar Questions
Explore conceptually related problems
Recommended Questions
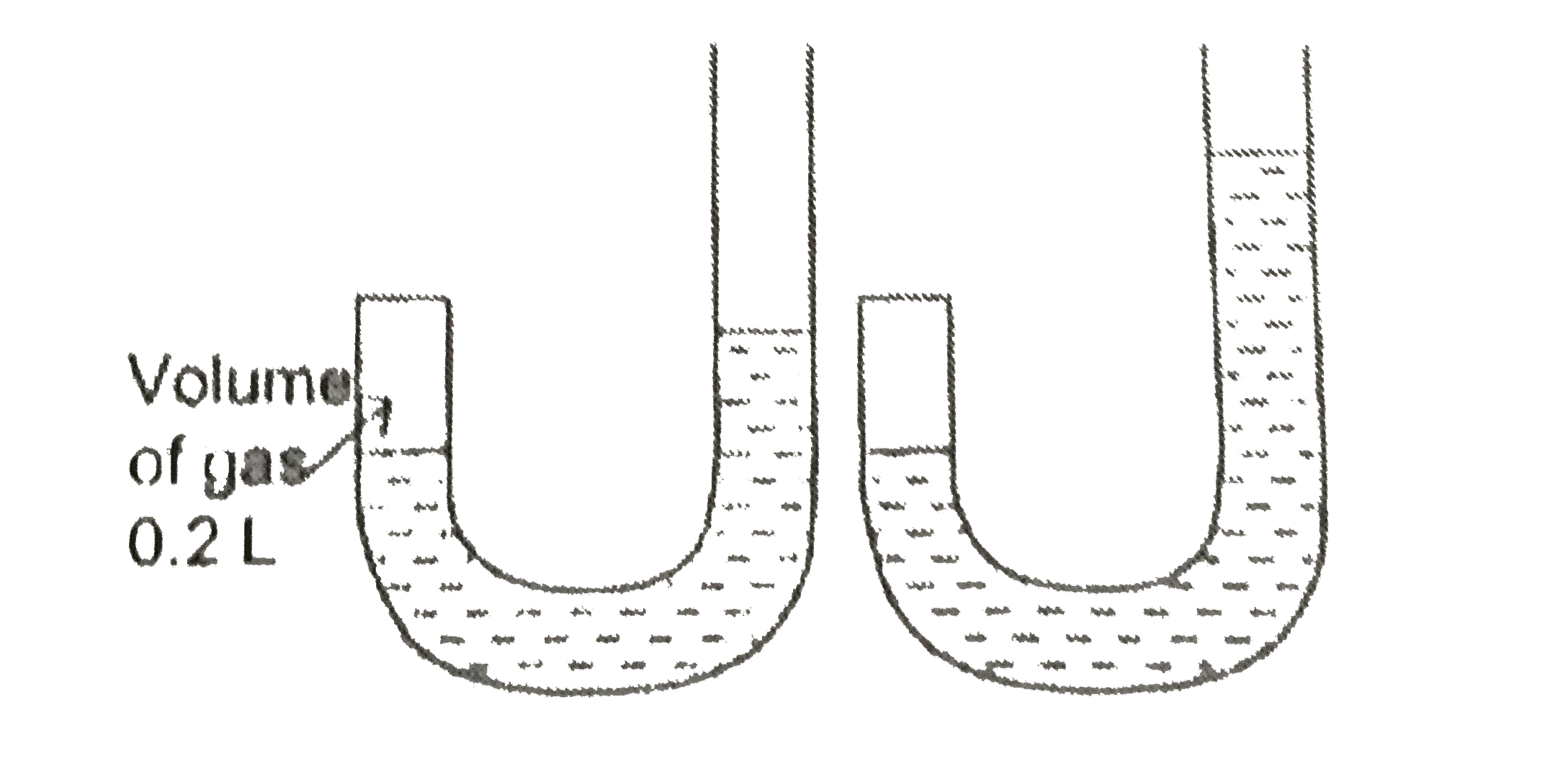
- J-shped tube closed at one end was used by Boyle to study the relation...
Text Solution
|
- A uniform tube closed at aone end, contains a pellet of mercury 10cm l...
Text Solution
|
- A quantity of gas is collected in a gradutated tube over the mercury. ...
Text Solution
|
- The narrow tube with one of its ends sealed as shown in the figure, is...
Text Solution
|
- An ideal gas is trapped between a mercury column and the closed lower ...
Text Solution
|
- J-shped tube closed at one end was used by Boyle to study the relation...
Text Solution
|
- The volume of a gas at 27^(@)C and 760 mm atmospheric pressure is 500 ...
Text Solution
|
- यदि किसी बर्तन में भरी गैस का दाब, वायुमण्डलीय दाब से 0.8 मिमी पारे के...
Text Solution
|
- The volume of a gas at NTP is 40 mL. If the pressure is increased to 8...
Text Solution
|