(a) Potassium dichromate is prepared from chromite (`FeCr_(2),O_(4)`) by fusing with molten alkali in the presence of air
`FeCr_(2)O_(4)+ 16NaOH+ 7O_(2) to 8Na_(2)CrO_(4)+ 2Fe_(2)O_(3)+ 8H_(2)O`
The solution of sodium chromate is filtered and acidified with dil , `H_(2)SO_(4)` giving sodium dichromate
`2NaCrO_(4)+ 2H^(+) to Na_(2)Cr_(2)O_(7) + 2Na^(+)+ H_(2)O`
Sodium dichromate is more soluble and less stable than potassium dichromate. Potassium dichromate is prepared by mixing a hot concentrated solution of `Na_(2)Cr_(2)O_(7)` and KCl in equimolar quantities.
`Na_(2)Cr_(2)O_(7)+ 2KCI to K_(2)Cr_(2)O_(7) + 2NaCl`
`K_(2)Cr_(2)O_(7)` is obtained as orange crystals,
Reactions (1) `K_(2)Cr_(2)O_(7)` oxidises ferrous sulphate to ferric sulphate.
`6FeSO_(4)+ K_(2)Cr_(2)O_(7)+ 7H_(2)SO_(4) to 3FeSO_(4)+ Cr_(2)(SO_(4))_(3)+ K_(2)SO_(4)+ 7H_(2)O`
OR `Cr_(2)O_(7)^(2-)+ 6Fe^(2+)+ 14H^(+) to 2Cr^(3+)+ 7H_(2)O+ 6Fe^(3+)`
(ii) `K_(2)Cr_(2)O_(7)+ 6KI+ 7H_2SO_(4)+ Cr_(2)(SO_(4))_(3)+ 7H_(2)O+ 3I_(2)`
OR `Cr_(2)O_(7)^(2-)+ 6I^(-)+ 14H^(+) to 2Cr^(3+)+ 3I_(2)+ 7H_(2)O`
(iii) `K_(2)Cr_(2)O_(7) + 14HCI to 2KCI+ 2CrCl_(3)+ 7H_(2)O+ 3Cl_(2)`
(iv) `K_(2)Cr_(2)O_(7)+ 4NaCl+ 6H_(2)SO_(4) to 2KHSO_(4)+ 4NaHSO_(4)+ 2CrO_(2)Cl_(2)+ 3H_(2)O`
(b) Potassium dichromate on heating with alkalies (KOH) changes to potassium chromate, which is yellow :
`K_(2)Cr_(2)O_(7)+ 2KOH to underset("(Yellow)")(underset("Pot. chromate")(2K_(2)CrO_(4)))+ H_(2)O`
On acidifying, the yellow colour change back to orange red due to reversible reaction.
`2K_(2)CrO_(4) + H_(2)SO_(4) to underset("(Orange)")(K_(2)Cr_(2)O_(7)) + K_(2)SO_(4) + H_(2)O`
The two main species involved are `CrO_(4)^(2-)` and `Cr_(2)O_(7)^(2-)` ions. Their structures are given below:
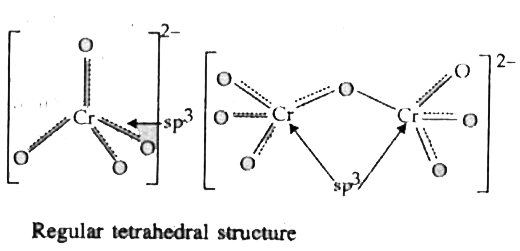
Regular tetrahedral structure
