Text Solution
Verified by Experts
Similar Questions
Explore conceptually related problems
Recommended Questions
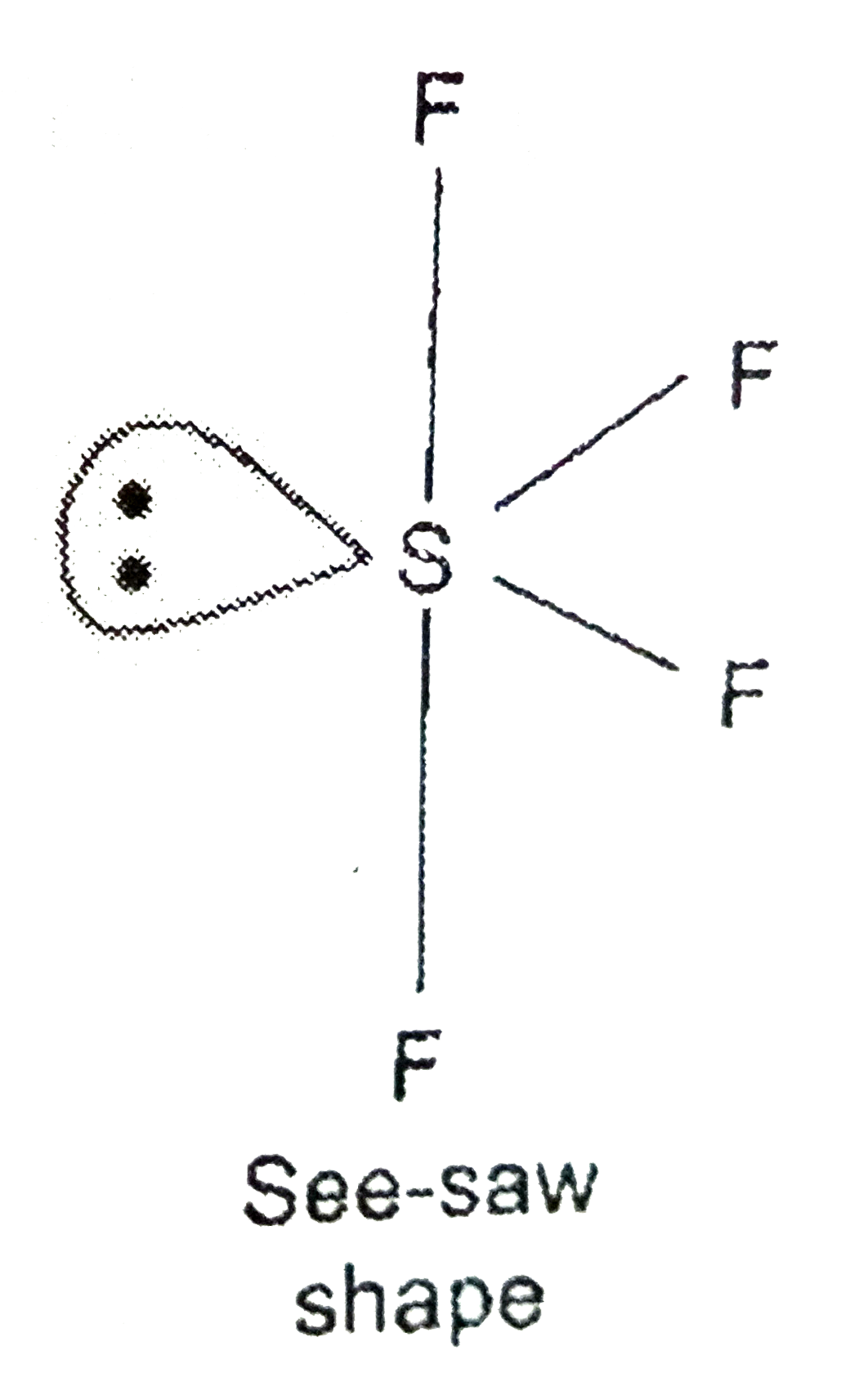
- Predict hybridisation and shape of SF(4) molecule.
Text Solution
|
- The hybridisation of sulphur in SF(4) is
Text Solution
|
- Predict hybridisation and shape of SF(4) molecule.
Text Solution
|
- Atomic orbitals involved in hybridisation of SF(6) molecule
Text Solution
|
- The atomic orbitals involved in hybridisation of SF(6) molecule is
Text Solution
|
- Predict the shapes and hybridisation of the following molecules CO(2).
Text Solution
|
- The hybridisation and shape of SF(6) is respectively
Text Solution
|
- Mention the shape of SF(6) molecule.
Text Solution
|
- The central S-atom of SF(6) molecule is hybridised.
Text Solution
|