Similar Questions
Explore conceptually related problems
Recommended Questions
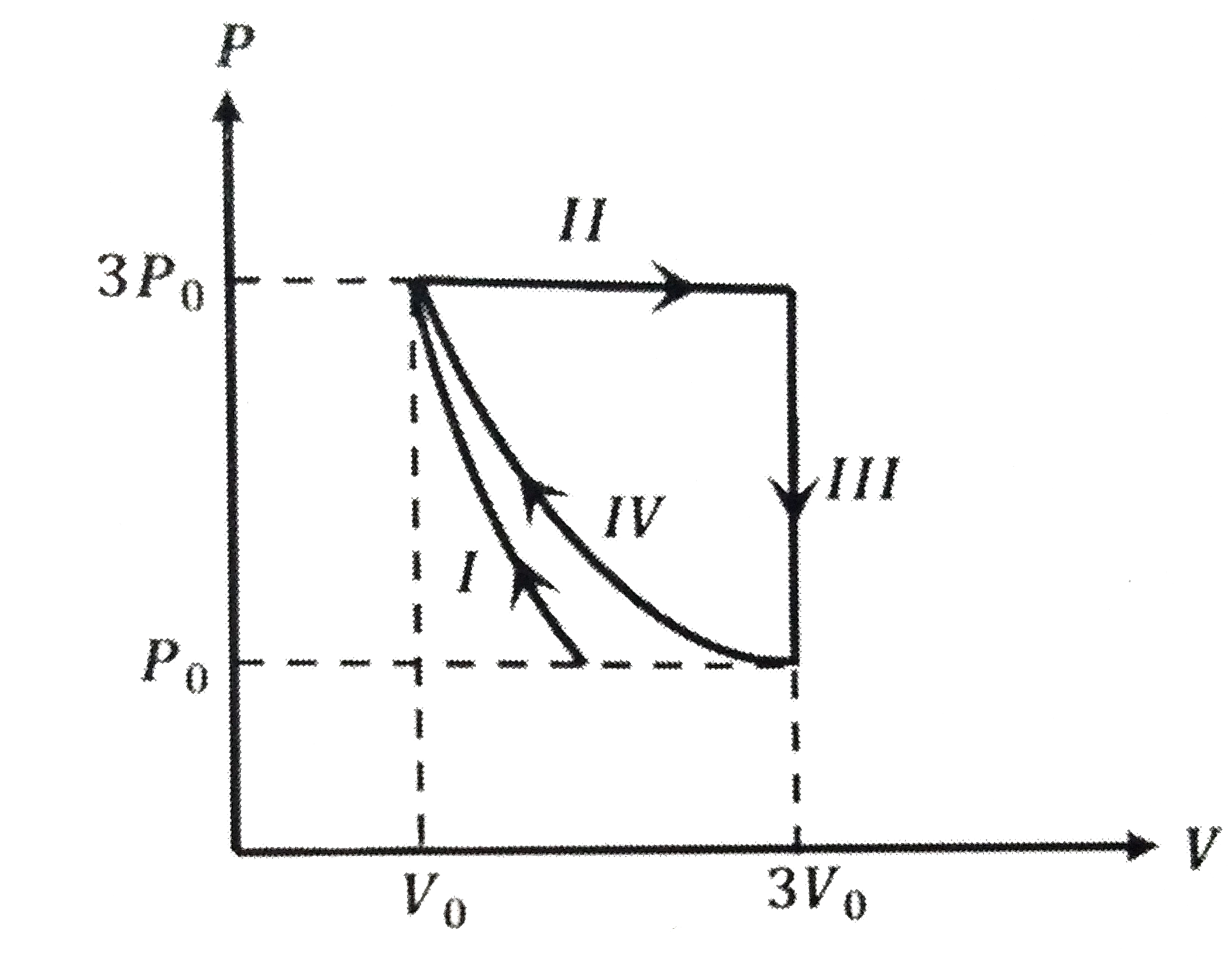
- One mole of a monatomic ideal gas undergoes four thermodynamic process...
Text Solution
|
- One mole of an ideal gas is carried through a thermodynamics cycle as ...
Text Solution
|
- An ideal gas undergoes for different processes from the same initial s...
Text Solution
|
- One mole of a monatomic ideal gas undergoes four thermodynamic process...
Text Solution
|
- An ideal gas undergoes for different processes from the same initial s...
Text Solution
|
- One mole of monatomic ideal gas undergoes the process ArarrB , as in t...
Text Solution
|
- एक परमाण्विक आदर्श गैस (monatomic ideal gas) का एक मोल (one mole), चार...
Text Solution
|
- एकपरमाणिवक आदर्श गैस का एक मोल चार उष्मागतीय प्रक्रमों से गुजरता है , ...
Text Solution
|
- An ideal gas undergoes four processes: isochoric, isobaric, isothermal...
Text Solution
|