A
B
C
D
Text Solution
Verified by Experts
The correct Answer is:
Topper's Solved these Questions
THE SOLID STATE
DISHA PUBLICATION|Exercise EXERCISE -1 : CONCEPT BUILDER|70 VideosTHE SOLID STATE
DISHA PUBLICATION|Exercise EXERCISE -2 : CONCEPT APPLICATOR|30 VideosTHE s-BLOCK ELEMENTS
DISHA PUBLICATION|Exercise Exercise - 2 : Concept Applicator|30 VideosTHERMODYNAMICS
DISHA PUBLICATION|Exercise Exercise|114 Videos
Similar Questions
Explore conceptually related problems
DISHA PUBLICATION-THE SOLID STATE-EXERCISE -2 : CONCEPT APPLICATOR
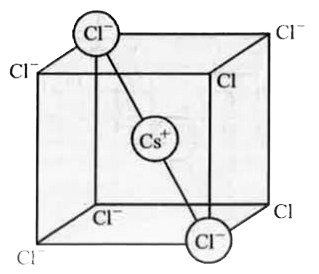
- CsClcrystallizes in body centred cubic lattice. If 'a' is its edge len...
Text Solution
|
- Non-stoichiometric compound have the properties of:
Text Solution
|
- The ability of a substance to exist in two or more crstaline forms kno...
Text Solution
|
- For a cubic geometry the limiting (r^+)/(r^-) is:
Text Solution
|
- The intermetallic compounds LiAg crystallises in cubic lattice in whic...
Text Solution
|
- A compound formed by elements A and B crystallizes in cubic structure,...
Text Solution
|
- A compound formed by elements X and Y crystallises in a cubic structur...
Text Solution
|
- Na and Mg crystallize in bcc- and fcc-type crystals, respectively, the...
Text Solution
|
- A match box exhibits
Text Solution
|
- Li' forms a body centered cubic lattice. If the lattice constant is 3....
Text Solution
|
- Coordination number of "C" in Be2C3 whose structure is correlated wit...
Text Solution
|
- The fraction of volume occupied by atoms in a primitive cubic unit cel...
Text Solution
|
- The fraction of volume occupied by atoms in a body centered cubic unit...
Text Solution
|
- The fraction of volume occupied by atoms in a face centered cubic unit...
Text Solution
|
- Moldydenum ("At. mass=96g//mol"^(-1)) crystallizes as bcc crystal. If ...
Text Solution
|
- The atomic fraction (d) of tin in bronze (fcc) with a density of 7717 ...
Text Solution
|
- Which one of the following has minimum value of cation/anion ratio ?
Text Solution
|
- What is the number of tetrahedral voids per atom in a crystal?
Text Solution
|
- Which set of following characteristics for ZnS crystal is correct?
Text Solution
|
- By X-ray diffraction it is found tht nickel ("at mass"=59g "mol"^(-1))...
Text Solution
|
- CsBr crystallises in a body centered cubic lattice. The unit cell leng...
Text Solution
|