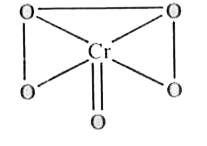
A
B
C
D
Text Solution
Verified by Experts
The correct Answer is:
Topper's Solved these Questions
THE D- AND F- BLOCK ELEMENTS
DISHA PUBLICATION|Exercise EXERCISE - 1 CONCEPT BUILDER (TOPIC 3: LANTHANOIDS AND ACTINOIDS)|20 VideosTHE D- AND F- BLOCK ELEMENTS
DISHA PUBLICATION|Exercise EXERCISE - 2 CONCEPT APPLICATOR|30 VideosTHE D- AND F- BLOCK ELEMENTS
DISHA PUBLICATION|Exercise EXERCISE - 1 CONCEPT BUILDER (TOPIC CHARACTERISTICS OF D-BLOCK ELEMENTS )|20 VideosSURFACE CHEMISTRY
DISHA PUBLICATION|Exercise Exercise-2 :Concept Applicator|30 VideosThe p-Block Elements (Group 13 and Group 14)
DISHA PUBLICATION|Exercise Exercise-2: Concept Applicator|30 Videos
Similar Questions
Explore conceptually related problems
DISHA PUBLICATION-THE D- AND F- BLOCK ELEMENTS -EXERCISE - 1 CONCEPT BUILDER (TOPIC 2 : COMPOUNDS OF TRANSITION METALS)
- The oxoanion in which the oxidation state of the central atom is same ...
Text Solution
|
- When an aqueous solution of copper (II) sulphate is saturated with amm...
Text Solution
|
- When concentrated HCI is added to an aqueous solution of CoCl(2...
Text Solution
|
- Colourless solutions of the following four salts are placed separately...
Text Solution
|
- The correct statements of the following
Text Solution
|
- Among the following, the compound that is both paramagnetic and colour...
Text Solution
|
- K(2)Cr(2)O(7) on heating with aqueous NaOH gives
Text Solution
|
- What would happen when a solution of potassium chromate is treated wit...
Text Solution
|
- The oxidation state of chromium in the final product formed by the rea...
Text Solution
|
- The final products formed on the addition of KI to copper sulphate sol...
Text Solution
|
- KMnO(4) (acidic / alkaline ) is not decolurised by
Text Solution
|
- Acidified K(2)Cr(2)O(7) solution turns green when NasO(3) is added to ...
Text Solution
|
- KMnO4 can be prepared fromK2MnO4 as per the reaction: The reaction ...
Text Solution
|
- The pair of compounds that can exist together is:
Text Solution
|
- In acidic medium, H(2)O(2) changes Cr(2)O(7)^(-2) to CrO(5) which has ...
Text Solution
|
- The reaction of aqueous KMnO(4) with H(2)O(2) i acidic conditions give...
Text Solution
|
- Which of the following processes does not involve oxidation of iron ?
Text Solution
|
- Assuming complete ionization , same moles of which of the following co...
Text Solution
|
- Which one of the following statements is correct when SO(2) is passed ...
Text Solution
|
- Name the gas that can readily decolourise acidified KMnO(4) solution:
Text Solution
|